Episode 263: Psychiatrist Effect in First-Episode Psychosis: HAMLETT Study, Antipsychotic Tapering, Dopamine Supersensitivity & Sex Differences with Franciska de Beer
Puder:
Welcome back to the podcast. Today, I'm thrilled to be speaking with Franciska de Beer, MSc. She's a talented researcher, soon to finish her PhD at the Center for Clinical Neuroscience and Cognition at the University Medical Center Groningen, Netherlands. Franciska is the first author of multiple groundbreaking articles from the HAMLETT-OPHELIA Consortium, including key publications in JAMA Psychiatry (Sommer et al., 2026; Sommer et al., 2026), World Psychiatry (Gangadin et al., 2025), Psychological Medicine (de Beer et al., 2025), and Schizophrenia Research (de Beer et al., 2024). I first reached out to her after stumbling upon her article that she first authored called “Psychiatrists effect on positive symptom severity and daily functioning during pharmacotherapy for first-episode psychosis patients” (de Beer et al., 2024b). Many of you will know I often cite McKay's study on psychiatrist effect and depression (McKay et al., 2006), one of the only psychiatrist effect studies. So I was overjoyed to find a second study that looked at psychosis and the psychiatrist effect. I've since learned more about her research interest and have been intrigued by her work on first-episode psychosis. And I think there's a kind of a new question of when we should continue antipsychotics, when we should not continue antipsychotics. And so hopefully we'll have some of the new research on that discussed in this episode. So welcome to the podcast.
de Beer:
Thank you for having me.
Why the Psychiatrist Matters: Psychiatrist Effect (01:35)
Puder:
So maybe we could start with the paper on psychiatrist effect and go from there. So do you want to describe the basic findings of this research?
de Beer:
Yeah, sure. I love your introduction and I think the McKay article, you love, I love it so much as well. So I think one of the main issues when we talk about psychiatry is that of course, we want to improve patients' recovery. Right? And the main way we go about it is look at, okay, how can we address patient characteristics like their economic wellbeing, like their social functioning? And we tend, I think, to overlook that it is someone else across the table that also has an influence in these patients' outcomes, namely the psychiatrists. And I think that is some literature or there is some decent literature about psychotherapy and the effect of a therapist in psychotherapy. But the person also matters. The clinician matters when there are drugs prescribed in pharmaceutical and pharmacological therapies. And that is what we wanted to address.
de Beer:
What is the effect of the psychiatrist when patients were recovered from first psychosis in that treatment outcomes? So what we did, is we have a nationwide study, the HAMLETT study (Begemann et al., 2020), that has about 350 patients, where we use slightly less of them from many different psychiatrists. And that allowed us to have 18 psychiatrists with an average 11 patients per psychiatrist. And so we could see, okay, does the patient's outcome over a period of 12 months, over one year, depend on the psychiatrist? And we indeed thought that that was the case. So we found that the psychiatrist could explain about 10% of the patient's outcome after one year, which is quite a decent amount. And what was quite interesting is that it's not only about the psychiatrist themselves, but also about how they prescribe medication, because we saw a significant interaction effect with the drugs they prescribed. And I think that is the new value of this study. We not only show that it's the psychiatrist, but also the way psychiatrists prescribe medication that matters.
Puder:
So the basic group of people, these were people that had first episode psychosis and then they were stable. Right? They had low scores on their psychosis scale. And so these were either randomized to be tapered or to stop the antipsychotic medication or to continue the antipsychotic. Right?
de Beer:
Exactly. Exactly. Yes. So during this study period of 12 months, they were initially randomized to either reduced antipsychotic medication or continue it for a period up to one year after remission. But in this study, our follow-up period extends that. So we started to follow them when they were half a year in the remission. So we had the trial phase, and we also had the phase afterwards, which gives us basically two views. One, a psychiatrist aiming to maintain a patient on either the treatment arm and the phase afterwards where psychiatrists were free to either reduce or increase, well, increasing always, but reducing the dose or, or not to.
Puder:
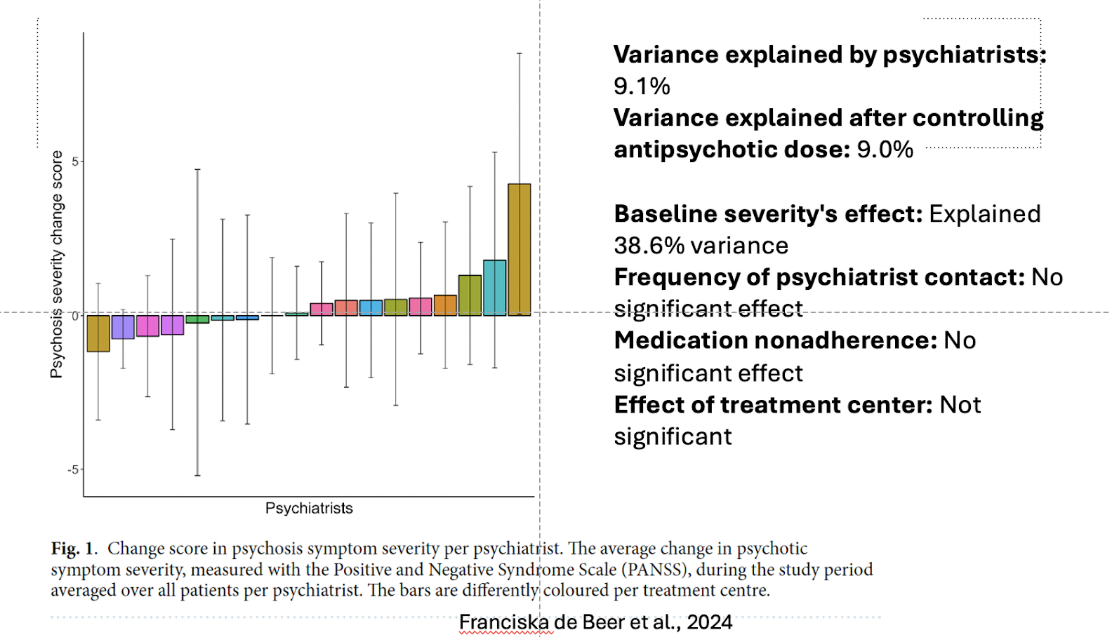
Great. So I've actually done a presentation on psychiatrist effect, reflective function therapist effect. And I'm going to pull up one of my slides. Maybe you can explain it. It's figure one from the paper. Let me just share this here real quick. For those of you who are on YouTube, you can see this [here]. So we'll try to describe it for those listening. Let's see here. Let's just play from this current slide. So describe for the audience, what we're seeing here in terms of psychosis severity change scores based on different psychiatrists.
Visualizing Psychiatrist Effect: PANSS Positive Symptom Change Scores Across Clinicians (05:53)
de Beer:
Yes. So what we see here is a bar plot where we see one bar per psychiatrist. And what's plotted on the y-axis is does a patient improve or worsen in their amount of psychotic symptoms. So what we did is we measured symptoms of severity with the PANSS, the positive and negative syndrome scale. The positive item scale: So, ”How many delusions do you have? How many hallucinations do you experience on a daily basis?” And while a researcher rates this scale, we did this shortly after they were in remission three to six months. And we did it again after one year. And basically what we did is how does one improve or worsen over the period of a year? And we averaged those improvements and decreases over all patients of a certain psychiatrist. So a psychiatrist had on average 11 patients, so you have an average of 11 patients.
de Beer:
What we see here is that some psychiatrists show a marked, well, a slight worsening, but not too different from zero. So basically the patients remain quite stable. Then there are also some bars indicating that some psychiatrists may actually show quite a marked improvement over the course of a year. And I think this bar plot actually shows, indicates that well, some psychiatrists, some patients with some psychiatrists have markedly improved outcomes compared to patients from other psychiatrists. This may be a bit controversial to say, but that is the belief that, well, there are some clinicians that may be more effective than others. Some literature suggest this may be 15 to 20% of psychiatrists having better outcomes and 15 to 20 maybe worse outcomes. I'm not sure about the percentages, but I do think that intuitively it makes sense that some psychiatrists may be more efficient in giving therapy than someone else. And their bar plot, I think quite effectively shows that
Puder:
The variance explained by psychiatrists was at 9.1%, and the variance explained after controlling for antipsychotic dose was 9%, which was interesting to me. Like the dose didn't change that some psychiatrists did a great job. Meaning that it's not purely dose-related that some psychiatrists did better than other psychiatrists.
de Beer:
Exactly. And I think the personal skills of a psychiatrist, the communication skills, are of massive importance. And I can't imagine, especially with psychosis patients, where there may be a tendency for paranoia. I mean, especially when someone [has a hard time trusting] anyone, I think it takes quite some dedication and some patience and massive communication skills to build this level of trust. So a patient's really open and able to explain and to be that honest about some symptoms, because it can be so personal and scary to explain to, I think especially with psychosis patients, the psychiatrist effect is of major importance.
Puder:
Yes. One of the things I've tried to convey to residents over the years is the amount of energy that we as psychiatrists spend on developing those skills, the developing our psychotherapy skills, needs to be as important as learning all of the details of neurotransmitters and different medications and side effects and management of those med side effects. One thing that was interesting, I'm wondering if we could kind of also mention here, is baseline severity explained 38.6% of the variants in this group. Tell me about that. That amount, it seems, it's about three, four times as much as the effect of the psychiatrist. So baseline severity was very potent in predicting how well someone was going to do. Tell me about that and what you think about that number.
de Beer:
Yeah, so it basically means that how well you recovered initially, predicts how well you will do after one year. So if you recovered really well, then the chances are that you'll be doing really well after a year are bigger, then when you recovered very poorly, because then your prognosis is somewhat bleak.
Puder:
Yes. And I think that is, it's a bigger impact than psychiatrist effect, baseline severity effect, that early response. Right? But there still is a psychiatrist effect, which is pronounced, which is also similar to what we see with therapist effect. It's around that 9%, which is interesting to me. Some things didn't have an effect at all. Right? Like the frequency of the psychiatrist contact, medication non-adherence, effective treatment center. How do you make sense of these things?
de Beer:
Yes. So I think the frequency of psychiatrists was overall fairly similar, so I'm not sure. We didn't have massive outliers. I think in the Netherlands, the frequency was fairly consistent overall, so there were no major differences. I can understand that. That was a contributing factor. And these patients are also embedded in a bigger care system where they do not only see psychiatrists, but also nurses and psychologists, and the frequency of seeing a psychologist was important for psychosis severity. So I think one issue that we did not address here, but would be lovely to address in future studies is that how does the team impact recovery? I mean, what kind of resources do you need for patients’ recovery? And also, who benefits from what? Because there may be a young male, early twenties, they may need different treatment needs than someone, for instance, women being vulnerable to psychotic episodes during perimenopause, where there are also other somatic complaints. So yeah, I would love to be looking at what the best combination would be, per person.
What Makes Some Psychiatrists More Effective? (12:16)
Puder:
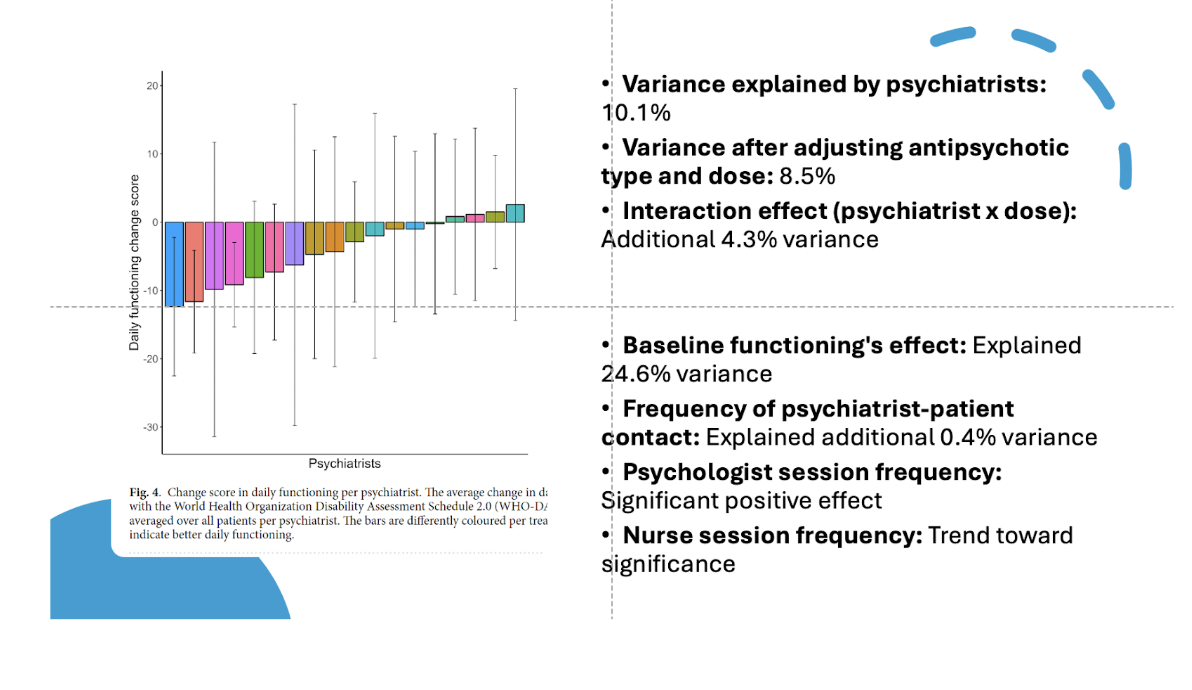
That's great. Okay. Let's go to the next slide here. So this is the, we're now going to be talking about daily functioning change scores. And so this is like another piece of the variance that they looked at and they found the variance of this was 10%. So tell me about this
de Beer:
As the next two, how many symptoms patients experienced in their daily lives. We also looked at how well they were able to get along with their daily functioning. You can think of how well they were able to communicate with other people during the household calls, whether they will be able to work or not. They rated their own daily functioning, and we saw that when, also similar to the psychosis severity data, patients with some psychiatrists showed better improvements and better improvements in this case means a greater reduction in symptoms severity in daily functioning. And this was 10%, which is quite a lot. And we also saw that there was a significant interaction with those, explaining 4%. And what that means, basically, is that it doesn't only matter who your psychiatrist is, but it also matters how they prescribe the antipsychotic drugs. And I think here, that is this combination of this interpersonal aspect, like how much trust is someone being able to build up with a patient, or how much openness is there about ongoing symptoms, and how well is a psychiatrist able to address these symptoms, for instance, because they emerge very suddenly. Or you need different kinds of treatments with fewer side effects. So that needs to be a switching or dose adjustments. And I think some psychiatrists may do this adjustment more efficiently than others.
Puder:
Yes. So, you know, we think about what wasn't measured here, we didn't really measure in this study why some psychiatrists were better than other psychiatrists, which has always been a question of mine. Therapeutic alliance of course, coming together with the patient trying to have consistent goals. What kind of things do you think might have been going on that made one psychiatrist better than another psychiatrist?
de Beer:
I think that's a very intriguing, difficult question to address. I think it truly has to do with the ability to build trust because of the tendency of paranoia. Then, also most likely, the degree to which decisions were openly discussed, like the shared decision making. And I think the process of shared decision making is crucial. For instance, when talking about medication adherence, I think it's so important that both parties are on the same page when a drug is prescribed. And even basically to choose which drug you want to choose, that should be ideally, if whenever possible, of course, I understand there may be cases it's not possible, whenever possible this should be an open conversation.
Puder:
Yes. I think a lot of the issues that I run into in my own private practice are things like anosognosia, where the patient has no insight into their illness. This can create increased difficulty in even getting the patient to agree to take a medication. Sometimes, I've started kind of a new approach with just doing pure psychotherapy without medication for the first couple months, just because the patient's refusing medication, but maybe not so flagrantly psychotic, but not to the point where they're easily hospitalized or put in a hospital, injected with an antipsychotic or something.
Any other thoughts on what kind of things, what insights this can give us as psychiatrists for what we could do to improve our ability as psychiatrists?
de Beer:
I think the illness insight is a very, very good one because where do we start if someone is not willing, and I think this is an extremely difficult one, but important one to address before starting on LAI, or perhaps more, admitting someone to a hospital. So I like your approach very much on first trying psychotherapy in the first case.
What Patients May Benefit from Antipsychotic Tapering? (16:44)
Puder:
How is it in the Netherlands? What is the approach for first episode psychosis? What normally happens to a patient? Are they admitted to the psychiatric hospital? Are they started on medications in the psychiatric hospital or they started outpatient?
de Beer:
That differs greatly on whether or not hospitalization is necessary. So, in many cases, there is a hospitalization. In those cases, they are often antipsychotic prescribed, say 10 milligrams olanzapine, is extremely common for patients to receive. I think that's the most common way. Also, in outpatient clinics, therapies are started. Typically, again, medication with some psychotherapy. I think those would be the most common routes. And it depends on whether someone has, whether the psychosis derails a lot, so hospitalization was necessary, or the person in the environment intervenes in time to go to a regular GP and start antipsychotics, either there, or with a specialized psychiatrist.
Puder:
I've heard about in Amsterdam, the rates of high use of highly potent THC. Does this play a factor in how you determine if you're going to try to take someone off of an antipsychotic or not? Have you, has there been any thought on that?
de Beer:
Well, I think that implicit, that was the assumption that when someone has a drug-induced psychosis and the person has really stopped using the antipsychotics, they may be the better suited persons to taper off antipsychotics. And interestingly, very recently, I have done a study on what happens after people stop antipsychotics. So patients tapered off their drugs, not all of them, but part of them in the HAMLETT study. And when it tapered, some of them actually reached those zeros that it completely discontinued. And in those patients, I looked, “Okay, but if they discontinue, what happens after that stopping point?” And we followed them up for about four years and we did a cluster analysis because we were interested. “Okay, what kind of trajectories do people typically follow when they stop the drugs?” What we found is that there were about two groups who started the antipsychotics fairly quickly again [pending publication].
de Beer:
So say after five months one group of these was also hospitalized fairly easily or quickly. So after four, five months, a subset of patients, 9%-- I still think 9% is not too many– but 9% of the patients who stopped, they needed hospitalization. And typically these were male patients with lower education and quite high care needs, so about double the care needs of other patients compared to others. Then there was also a group of patients who were able to stop medication for a very long time, did not relapse, did not have hospitalization. And those were people who were fairly young. The first psychosis, they had good education, quite high functioning, the highest occupational rates, had partners quite often. And what we noticed, what we did not expect at all, was that they had initially quite high drug use. So we think these, a part of these patients who were able to stop medication fairly well, were those who had initial drug use, but then most likely stopped drugs, or at least way less, than initially. And those may be the patients that could do better without medication.
Short-Term Relapse Risk and Quality of Life with Early Tapering After First Episode Psychosis (20:22)
Puder:
Okay. That's great. That's good to hear. Let's talk about the JAMA article. This is, I'm just going to pull this up so people can see what this looks like. It says, “Early dose reduction or discontinue verse maintenance antipsychotics after first psychotic episode remission” (Sommer et al., 2026). So it looks like you were the second author on this. Congratulations. This is, I mean, you've done some great articles at such an early time in your career. Talk to us about this study. What was found? The main findings. Things that add to our discussion.
de Beer:
So for people's background, what we did is we randomized about 350 people who experienced a psychotic episode for the very first time. So, the first episode psychosis. And they were randomized to either dose reduced antipsychotics faster than guidelines prescribed, so within the first year after remission, or they could have to maintain the dose in which they reached remission for at least one year. That is because there's quite a debate about what would be best. I mean, can we taper off earlier? We know that it has a risk of increased relapse, but I mean it may have long-term functional benefits. And in fact, there was a Dutch study that showed that in the long term, after seven years, the group who dose reduced early had long term functional benefits (Wunderink et al., 2013). And that caused, at least in the Netherlands, quite some fear with some psychiatrists dose reducing early, others maintaining to the guidelines more.
de Beer:
And while, with that, there was quite some difference between how psychiatrists dealt with the situation. So there was actually a need for a big randomized-control trial to give some more solid, at least in the bigger group, evidence. So what we saw after we looked, “Okay, so what happens one year after people were in remission?” and we saw that those whose dose reduced early had a higher risk of relapse compared to those who maintained stable on the dose. And also that the dose reduction group had a lower quality of life compared to the maintenance group. And now there's something I think is very important to say here, that this [lower quality of life] is only true for the male patients.
de Beer:
The quality of life reduced in the dose reduction group in male patients, not in the female patients. I think that is fairly interesting. And it caused the question of, “Okay, but were the female patients dose too high initially then, or are they better able to dose reduce?” I mean, does it have something to do with potentially larger social groups or that they are more open about their symptoms? So we do not know the exact courses, but I think this is a very relevant finding to mention here. Also, this was short term. Now, we were also interested, of course, in what happens in the longer term because we knew that dose reducing early has a greater risk of relapse. And what we saw after three or four years is that patients who dose reduced had better daily functioning compared to those who maintained that dose [and this better functioning is even stronger in women, so women have more benefits of early tapering too].
de Beer:
And now, what is crucial here is that this is not caused by a lower dose because at three and four years, they have fairly similar doses. Actually, the dose reduction group is [seems to be even though not significantly different] even slightly higher dose than the maintenance group. So what we see here is that there's functional benefit of early dose reduction, but that is not because of a lower dose, because the doses are the same. So it has to be something different. And I think we come back, maybe we touch upon the psychiatrist effect here. So does it? We think that the process of early dose reduction will have benefits, but then what is it about this process that could be beneficial? I mean, is it the educational component? Is it that a patient feels autonomy? Because when someone, a patient and a psychiatrist, they agree on, “Okay, let's dose reduce,” they do this together, extremely, extremely slowly.
The Art of Tapering: Shared Decision-Making and Patient Autonomy (24:48)
de Beer:
So dose reduction is a process of months. And that is a conversation, ongoing conversations between a psychiatrist and a patient. “Okay, how are you feeling? Can we go lower? Should we stabilize? Should we increase where the patient may also feel increased autonomy of, “I report these symptoms and now I can also sort of not completely make my own decision, but be involved in this process of medication decisions.” Also, they experience what happens to them when they lower the dose. I mean, they experience perhaps that when on a lower dose they have more symptoms. “And I feel like, okay, but this is then what this drug does to me and it helps me to not hear voices,” for instance, “and improve or at least keep my job,” or something. I think that is a learning component, as well. And I think that we have a slight backup from literature here because there has been a previous study where it said there have been two previous studies that have contradicting results. One said that long term early dose reduction is beneficial. And that one was open label, so both patients and psychiatrists knew what was happening (Wunderink et al., 2013). There's also one that was double-blinded. So both the psychiatrist and the patients did not know whether there was dose reduction or whether there was continuation. And that study, interestingly did not show a beneficial effect of early dose reduction (initial study: Chen et al., 2010;10 year follow up: Hui et al., 2018). And I think what that shows is that this open label process, so both patients and psychiatrists know what is happening and make deliberate decisions, it's crucial for this long-term benefit.
Puder (00:29:07):
You know, I'm also thinking about how, if you're a psychiatrist and outpatient yet, and this is kind of what you were saying, but maybe I'll just reiterate it one more time, it's like if I reduce the dose and let's say the patient is skeptical that they need to be on this at all. Right? So I reduce the dose and they see that some symptoms come back and it's enough symptoms to let them know that the symptoms are back and they're distressed, but it's not so much that their insight is completely gone. Right? Because if all of the medication is taken away, then their insight can be completely gone. You, as a psychiatrist, are now part of their paranoid structure. Maybe you're trying to poison them. Maybe you're part of the CIA, the FBI, coming after them. So if all of their insight is stripped away completely it could be bad and some patients will stop their meds anyways. But if instead, your psychiatrist will be like, “No, let's reduce. We're going to talk. We're going to see if it's worse, better.” And then they come back and they can talk and you then you have more time to gain that rapport, as well. Before, they just quit altogether. Because I've had patients who are like, “No, absolutely not. Don't want to be on anymore. Never want to come back.” Right? And then struggle.
de Beer:
Yes. And I think that's a crucial aspect here, is that it does really matter that there's good monitoring. So when you dose reduce, there should be really regular monitoring. I mean, ideally every week, but very, very frequently, at least. And this could be done by the psychiatrist, but in the Netherlands it's also done by nurses and psychologists. So the workload is slightly well distributed among people. And I think this is crucial because if you don't have caregivers who know what, I mean, everyone has these relapse symptoms which are quite personal, like the stress, the sleep, is it hearing voices more often? Is it a specific type of voice? Everyone will have their own landmarks for relapse. And I think having a caregiver that knows them personally and is able to monitor them very frequently, that is crucial because we should not just discontinue or dose reduce without any guidance. I think that's the worst idea ever. So we need to be able to provide very good monitoring.
Dopamine Supersensitivity: How Tapering Differs by Antipsychotic Class and D2 Receptor Affinity (28:52)
Puder:
Yes. Yes. Very good. Okay. And then there was a letter that you guys did that was kind of commenting on someone else's critique of the study or something. I'm curious. I'm just going to pull that up to show you what I'm talking about. But what came from this letter, what I was thinking about it, was we, you really differentiate between three types of antipsychotics and how they may differentiate in the long-term outcome of someone who's coming off of it (de Beer et al., 2025). Like if they're on a strong dopamine blocker like Haldol [haloperidol], risperidone; versus weak D2 antagonist, like olanzapine, quetiapine; or a partial D2 agonist like aripiprazole. So tell me about how these different medications differ when we are trying to bring someone off and more about the study (Gangadin et al., 2025). I guess the HAMLETT study that you guys were doing.
de Beer:
I think for a very long time people have sort of assumed that it's important what dose you prescribe and that when you taper off antipsychotic, it doesn't really matter what type of antipsychotic it is, but you're more into, “How much dose reduction is it?” And then the dose reduction would be sort of similar among all antipsychotics. But when you think about it, antipsychotics work differently on the dopamine system. There are quite some differences. There are these antagonists. Right? They bind to dopamine D2 receptors and they actively antagonize or they inhibit the dopamine signaling on that receptor. And you have antipsychotics, the antagonists who do this very heavily. So they bind very tightly and they block a lot. We call these the high dopamine D2 affinity antagonists and like haloperidol and risperidone. And there are low affinity antagonists. They also bind, but less tightly.
de Beer:
But, if they bind, they inhibit. These are olanzapine and clozapine; and we only have one, clozapine. So we categorize clozapine as in this group, too. But we only had very, very few cases of clozapine used in the HAMLETT study because, well these were remitted patients and typically, in the Netherlands, you give clozapine to treatment resistance. And then there's a different group of antipsychotics that, well they bind with very high affinity to dopamine receptors, meaning that you need a small dose to bind to many of the dopamine receptors in the brain. But when they bind, there is agonist, meaning there's stimulation, partial stimulation. So there is stimulation, but less than endogenous dopamine would stimulate the receptor. Meaning that, in the end, you have a reduction of your dopamine signaling because of antipsychotics, they all reduce dopamine signaling in one way or another. So that gives us, with the three types of antipsychotic drugs and what we were interested in. Okay, but does it matter if when we taper, what kind of drug we are tapering?
de Beer:
Because, on the one hand, there's this view that, “Okay, the speed by which you taper is really important and if you taper really fast, then that will give you the highest risk of relapse.” And we were like, “Yes, that will be very important, but it may also be of interest what type of antipsychotic you are tapering.” So we looked at, “Okay, does it matter how fast people are tapering?” So we compute, we have their medication trajectories. And so we could say, “Okay, they are starting tapering here and they ending it here. What is sort of the average peak they had over this trajectory?” And we calculated the tapering speed in milligrams a day, or in equivalents. And what we saw is that it is not the speed by which patients taper and antipsychotics. Well, and I have to note, none of them stopped abruptly.
de Beer:
They all did gradual tapering. So this is the difference between gradual tapering and even slower tapering. That's the difference. And then there was no difference as long as you taper gradually. Tapering even slower does not further reduce the relapse risk. But what is really important is the type of drug that you are tapering. So we saw that patients who used these high affinity antagonists, so with the high blocking dopamine blocking receptor antagonists, they had the highest risk of relapse. But they also had a faster risk of relapse. Meaning, when they stop the drug they relapse faster than patients on the low affinity receptors and partial agonists. And this categorization links to the idea of what we call dopamine super sensitivity, which is the idea that if you block dopamine receptors for a prolonged period of time, the brain will respond to this blocking by upregulating dopamine receptors.
de Beer:
And by regulating, we mean, the brain will make more dopamine receptors that are active and that can respond to dopamine. And so that would mean that if you block dopamine receptors stronger, there may be stronger upregulation. So we have categorized stronger upregulation with the high affinity antagonist and the least dopamine upregulation with partial agonist. Because with a partial agonist, there is still some dopamine signaling, so maybe less need for the brain to have more active dopamine receptors. And I think that because we saw that these high antagonists led to more relapses and faster relapses, that was an indication for this dopamine super sensitivity, which we then also assessed in our PET study.
Puder:
I was going to bring this up by the way. This is incredible. This is, I mean, there may be a lot of mental health professionals who are hearing this for the first time. And if this is at all confusing guys, please go on to my website, psychiatrypodcast.com. I'll have all the links to all these articles and we'll have a transcript up, as well. If something's confusing here, because I think this is, the first time you're hit with this information, it could be a little bit confusing. So I was looking at this PET scan and if you're watching on YouTube, you'll see the PDF and the PET scan showing these different brightnesses based off of the controls, the partial agonist versus the antagonist. So walk me through what this PET scan study found here.
de Beer:
Yes, so what we did is we wanted to know, “Is dopamine super sensitivity, do we see more dopamine receptors when people have used antagonist antipsychotics?” But the problem is you do not, you cannot measure this when people are on antipsychotics because, well, there's antipsychotics binding on the dopamine receptor, so you can't measure them. So what we did is we had people taper off antipsychotics and as soon as they really completely stopped, one week afterwards they were scanned in a PET scan, which stands for positron emission tomography, meaning they get a radioactive tracer injected and this radioactive tracer, called raclopride, binds to dopamine D2 and D3 receptors. Basically, you could see, like a Christmas tree with Christmas lights, and whenever there's a blob, you see a light in the Christmas tree, it means, “Ah, there's the receptor.” So what we did is we had them scanned one week after they stopped it, the medication, and two months later, and then we differed, of course, between the antagonizing versus agonizing drug.
de Beer:
So is it blocking the receptor and the partial agonist or is it partially stimulating the receptor? And we had a group of healthy controls that were comparable in age and sex and so on. And what we saw is that when people had stopped the antagonists blocking receptors, those have more dopamine receptors available shortly after they stop the medication compared to the healthy controls. Meaning, that indeed, these end antipsychotics, they upregulate the dopamine receptors. But what's very interesting, is that they pass your partial agonists, so when patients had used drugs that still simulated the dopamine receptor somewhat, they did not show this upregulation shortly after they stopped it.
Puder:
That is very interesting.
de Beer:
We do not know how and why, but what is crucial, I think to mention here, is that the antagonist group, 80% of them had a relapse after they stopped. Well, in the partial agonist group, it was only one person. Again, it's difficult to make any cause or inferences of course here, but it gives us a hint like, “Okay, maybe this dopamine dysregulation associated with antagonist is indeed related to relapse and that we do not see this upregulation shortly after people stopped partial agonists.” Yeah, maybe it's beneficial. We do not know. I think this should be really further investigated with future studies, but it gives us a hint.
Puder:
Yes. It gives us a hint and it makes sense to me. Imagine the dopamine receptor, like a door, and you are blocking the door with this very strong medication. And so, your brain is like, “Okay, these doors are being blocked, I'm going to need to make more doors.” And so that's what the strong antagonists, like how the risperidol do. That being said, in clinical practice, sometimes you need those medications initially. Or they can be beneficial to get a quick response or to help people. So I'm not, we're not going “all bad” on these things, but just to know, as clinicians, that sometimes this can then upregulate the amount of doors there are. Right? And so that's what you're seeing in this PET scan, very, very well, is there's more doors after you stop the medication. There's more dopamine receptors and that could cause a potential rebound issue.
de Beer:
Yes. And I'm very curious to know, so these were all first episode psychosis patients. I think this is also perhaps exclusive to first episode psychosis patients. So I'm not sure whether it would make sense, so for instance, what we don't know is okay, what if you need an antagonist? Well, you might start with the olanzapine instead of risperidone and haloperidol. But what would be the effect of adding aripiprazole? I mean, would that mitigate the effect? I think we have no idea whether that might be an option.
Puder:
I tend to not try to combine those because I know Abilify [aripiprazole] binds actually tighter, aripiprazole binds tighter than Haldol [haloperidol] or risperidone. So kind of it's just, you know, using two antipsychotics, for me, is not ideal. I would rather personally optimize one, get the blood level, the highest possible, see if it works. If it doesn't work, move to another medication. Okay, one of the interesting findings from this though was that long term, what happens to the PET scans?
de Beer:
Yes. So long term, we saw that the antagonist group, they still had an elevation, but it was not significant anymore from the difference from the health and controls. We do need to note here that quite a lot of the patients from the antagonist group, they dropped out because they had a relapse within two months. So our group sort of was cut into two. I think we may have a power issue there with finding a significant result because so many had a relapse. But then, in the past, our agonist group, well, except for one, they all made it to the second scan. And what we saw is that there's an increase in dopamine receptor availability, so without potentially more dopamine receptors. And we do not exactly know why and how because what could it be? Could it be an upregulation? Could it be upregulation in the temporal lobe, or not? We do not know.
de Beer:
I think it's something extremely interesting we should be looking into.
Speed of Tapering (41:28)
Puder:
I think that the group that did not relapse, the brain, over time, normalizes somewhat. Right? It was like, “Okay, we needed more doors when there was this blockade. Now we don't need as many doors.” And I think this is a general thought that I've had when treating patients, is that your brain slowly goes back to normal. When I'm tapering someone off of whatever it is, an SSRI or whatever medication, it may take time for the brain to adjust. And so a slow taper makes more sense. So you don't get a rebound issue. Is that, maybe it's too early to say that definitively, but I like that you guys are still….
de Beer:
Yes, and I think I like the gradual tapering both because it makes sense from a neurological point of view, but also because it gives you time to intervene in case symptoms emerge. I think for many patients finding the lowest effective dose can be eventually the end goal. So for some, they may want to start off with, “Okay, let's see if we can go to dose zero.” They may end up finding that that is not feasible, not possible. And that finding the lowest possible dose is the best option. And in that case, doing a really long, long, long, very gradually, is crucial because only after a few weeks your brain will have been able to adapt to this new dose and whether or not there will be symptoms.
Puder:
Well, when you talk in this study, the prior study, the HAMLETT study, “gradual versus ultra gradual dose reductions,” what was the time difference between those things? Like, give me an example with olanzapine 10 milligrams. Let's say they're starting with 10.
de Beer:
Well, the average was four months.
Puder:
Was that the normal tapering, four months?
de Beer:
That was about the average. And then they started off from a dose of I think about 10 mg day of olanzapine equivalents a day. And then they tapered off a course of three to four months. And I think we compared it to even whether a person was tapering slower than this or faster than, within two or three months. But they never tapered off within two weeks. That was our cut. We excluded all patients who tapered faster than that.
Puder:
And then the ultra rapid or ultra slow tapering, how long was that?
de Beer:
So that was longer than the three to four months. And I think we have a range up until a year. I think that's quite extreme, but I think in that group it will be more around six months.
Puder:
Okay. So four months, that's about 2.5 milligrams per month. And was it just a steady decrease or did some people drop it faster at first and slower in the end?
de Beer:
We also provided some tapering schedules from the HAMLETT study, which can also be found online; and there's a bit of a hyperbolic tapering. So initially there's some larger steps and in the end the steps are somewhat smaller. And the idea behind this is that it matters how much of the, how many of the dopamine receptors are pulled off from antipsychotics. And with many antipsychotics, you read a therapeutic window fairly easily. So say you read the idea behind a therapeutic window is that you should occupy about 60 to 80% of the dopamine receptors. And then that's the sort of the sweet spot. It's therapeutically effective, you have enough dose to be effective, but it's not too many receptors occupied. With high doses, if you move, for instance, from 10 to 5 milligrams of olanzapine, that makes a difference in how many receptors you occupy. But not so much in the final steps, because then it goes really fast. So we would recommend slightly earlier, slightly large steps and then in the end be extremely careful. We think the end is a very important part, where we should be extremely careful.
Sex Differences and Effects of Menopause on Antipsychotic Treatment (45:47)
Puder:
You talked about how it's different between men and women. Can you talk more about that and specifically any recommendations for women that you've been finding? I know also you've looked at women in different life trajectories, like menopause, and stuff like that. Tell me about that research that you found (de Beer et al., 2025b).
de Beer:
Yes. So for women, we found that it does not only matter whether it’s a woman or a man, I mean that differs quite a lot in how many, how much of an antipsychotic they should get. But it's also important what hormonal life phase they are experiencing. So, I mean, regarding type of antipsychotics, be careful with risperidone. I mean, it causes hyperprolactinemia and it may cause breast cancer (Taipale et al., 2024) So that is a big warning. And if risperidone, paired with aripiprazole because aripiprazole is able to decrease hyperprolactinemia. So that might, regarding the type of antipsychotic. So something else we've been doing is, so women on average, so the problem with antipsychotic prescribing guidelines, at least in the Netherlands, is that they do not differentiate between men and women. So men, women, doesn't matter.
de Beer:
They are prescribed similar doses. Given that women are on average smaller, they have smaller bodies, they have smaller organs, they have faster blood flow. So if you give a drug, the blood circulates faster. So medication can reach the brain faster. Intuitively, then, it would make sense to have different prescription guidelines. Still there are none, which it's maybe suboptimal. So what we did is that we looked at, “Okay, we have University Medical Center Groningen. We have many people who draw blood. Let's see if female patients differ from male patients and how much antipsychotic they have present in their blood. Let's see how old they were.” Because when people enter menopause, we know that estrogen drops and estrogen is important for how fast a drug is processed by the body. So estrogen is important, especially for olanzapine and clozapine because those drugs when you have high estrogens those are metabolized slower.
de Beer:
So there will be more of the drug present, hypothetically. But then, if you enter menopause, the perimenopausal transition, the estrogen drops, and while the drugs may be processed faster. And what we saw in this study is that there was no difference between men and women if they used aripiprazole or quetiapine, they had fairly similar blood levels (de Beer et al., 2026). But there was a difference for olanzapine and clozapine, where women had higher blood levels compared to men, but only in the ages before menopause. There, we saw the biggest difference. So when women are in the reproductive years, and they have higher estrogen levels, they also have higher levels of olanzapine and clozapine compared to men. There's not really any reason why women should have higher blood levels of antipsychotics compared to men. Because we know they also report high side effect burden compared to men.
de Beer:
What was interesting is that as soon as women enter menopausal ages, we see that the levels decrease to levels that are comparable to men. And so we were really interested in this like, “Okay, women have shown decreasing levels of antipsychotics when they enter menopausal ages. Can we have a deep dive into this? Because not every woman is the same. They will have different trajectories.” So we did a follow up analysis in which we, with machine learning models, made different trajectories of men and women of the blood concentrations over time because clozapine is monitored, at least in the Netherlands, quite regularly. So we know we have yearly measurements of blood per person for patients, and we know women undergo menopause transition roughly between the ages of 40 to 55.
de Beer:
So we had a look, and we classified the trajectories. And what we show is that most women, they show a decrease of clozapine levels between the ages of 40 to 60. And I think this is while men mostly remain stable. And that is extremely important because we know that clozapine is prescribed to treatment-resistant schizophrenia. So this is actually a very vulnerable group of women who show reduced levels of clozapine over time. And I think we should be very, very wary of this because these are women who are in clinical practices. I mean, they are monitored, they are seeing a therapist and they show reduced levels. They may be incredibly vulnerable for having a relapse compared, especially compared, to men. And this showed us that when women enter the menopausal ages, we should really be on the lookout for their levels and their symptoms. We should be monitoring them very closely.
Puder:
Yes. I just want to pull up Figure 2 here, to show people that can see this online. And you could see with men, it's very stable. The estimated mean clozapine concentration. Whereas, with women, it really does decrease. And we're talking about from around like a little bit above 500 to below 450 (de Beer et al., 2025b). And we know that 450 may be that place where they get that dose response, 350 to 450, somewhere in there. So one, I think, practical consideration from reading this paper and thinking about this would be to, if you do have a patient that's stable on clozapine, who's female who's around 40, 45, you want to get their blood level. You probably will have their blood level, but you want to get it if you don't, and then you want to adjust the dose as they get older to keep that blood level where that sweet spot is, where they are stable. And, if you have a patient that is not doing as well, this may be the reason why. As they go through menopause, if their estrogen drops, and then they're not doing as well on the clozapine dose that they've been doing well for years. It's counterintuitive to think that the clozapine level would drop as people get older. Because usually medication levels increase as people get older. So this is a good clinical pearl here.
de Beer:
Wonderful. Yes, absolutely.
Puder:
Wonderful. Yes. And then tell me about your PhD thesis. What are some of the things, if there's anything that we haven't talked about? Is there anything extra you want to kind of mention?
de Beer:
Well, I think the main message is that ideally we try to personalize the treatments very much to the patient. And then, I think that means that this is an interpersonal process. Like giving pharmacotherapy is an interpersonal and social process. That is something we should be acknowledging, I think, very explicitly. And we should try to understand why some psychiatrists are perhaps more effective than others and are they doing something that we can teach to other psychiatrists. At the same time, we should be very aware of what kind of a drug a patient is using and how the drug influences dopamine. And we should treat women absolutely differently from men. And when we treat women differently, we should be very mindful of the hormonal lipase. I think those are the main takeaway messages of my thesis.
Puder:
Great. It's been wonderful having you on. It's been great to kind of look through some of these studies. I hope that people listening go back and read them and get curious about them. If there's something, if you didn't understand, or something you want to look at, I will have a transcript on my website, psychiatrypodcast.com, which I have a team that goes through it just to make sure the wording is correct and we try to smooth things out where possible, so it's more readable, and we'll have all the citations there. But yeah, any final thoughts? Any final things that are still on your mind that you want to mention?
de Beer:
I think we covered a lot already. I'm very curious to see what the clinical implications of these are.
Reflective Functioning and How It Influences Therapist/Psychiatrist Effectiveness (54:28)
Puder:
Yes, I think this is great. Have you read the study on reflective function and therapist effect [Cologon, 2017]? Have you looked at that one yet? Okay, I'm going to have to show you this slide here. So there was a study, so I've been curious about therapist effect for a while. Okay. And what, initially the best study that I found on therapist effect was this study [Anderson, 2009] where they videotaped therapists and they had the therapists…. Let's see, this is it. They videotaped therapists responding to difficult interpersonal questions from actors. So they had clips and then they rated the therapist's response to these different, difficult, interpersonal situations that come up. Things like when a patient may say something like, “You can't help me.” Or, “I don't know what to talk about.” Or, “Only the therapist's opinion matters.”
Puder:
You know, like different conflicting things that the patient can say. So they videotaped the therapist and they looked at the scoring and they looked at how that scoring related to the therapist outcomes on the OQ-45, on a bunch of the patients they were treating. And they found a correlation of 0.47. It's somewhat a pretty good predictor. One of the best that I found. But then I stumbled upon this study called Therapist reflective functioning, therapist attachment style and therapist effectiveness, by John Cologon (Cologon et al., 2017), I'm still trying to get him on my podcast. So if anyone knows John Cologon, tell him to respond to my emails. So here, we had 25 therapists seeing 1,001 patients and they did OQ-45. But on this, they did the adult attachment interview (AAI) and they scored it with reflective function.
Puder:
Okay. And so, reflective function is the ability to mentalize your own emotions, your parents' emotions. What was going on the why of your childhood. Okay. And so, then they looked at the adult attachment interview, the reflective function score, which goes from negative one to nine, nine being the highest. And they found, they broke the therapists into three groups. Low reflective function score therapists had no change in the OQ-45 at all. So patients came in with an OQ-45 of 80, they left with an 80 at session 10. It's like the patients who had medium reflective function had scores that dropped from around 80 to, let's see, this is around 73. Okay. And the patients with high reflective, the therapists with high reflective function scores had to drop from 80 to 67.
Puder:
So, they looked at the variance of the therapist effectiveness and they found a 70.5% variance was accounted for by reflective function, which when I saw that I was like, “This is huge!” That's a huge number, right? 70.5%. So that took me on a journey to investigate reflective function. I've done a bunch of episodes on this [see episodes 213, 219, 249, and 260]. I won't belabor it. I'm actually releasing an episode [see episode 260] before our episode, with Miriam Steele, who's one of the original authors of reflective function. And I lead cohorts that try to increase provider reflective function. And so, in the future we may be doing research on that as well. But it's something to think about as there's therapeutic alliance, there's the ability to connect with patients, but I think there's also the therapist’s reflectiveness, which is based on the adult attachment interview.
de Beer:
So what do you think is important about the reflective ability of psychiatrists that is so influential?
Puder:
So when a difficult interpersonal situation occurs with a patient, it stirs up our own stuff. Right? And I think that people with high reflective function have done the work to understand their own countertransference, their own reaction to patients, to be able to put words to it in their own mind. And then, to be able to not necessarily get emotionally evoked in a way that would negatively impact the connectedness between you and the client. I think someone with a high ability to reflect on early attachment stuff, it hits the provider in an area that they can't fake. So the adult attachment interview, it's impossible to study for it, like you would a normal test. You go in there, if you try to have high reflectiveness, you will be found out, right there. There's no way of hiding because you've either done years of your own personal therapy that would allow you to have higher reflectiveness or you haven't, essentially.
How Therapists and Psychiatrists Can Improve Reflective Functioning (58:08)
de Beer:
And so do you think this is something that's innate that some people just do better than others? Or do you think this is something we can train with therapies? Because I think one part of the beauty of treatments and therapist effect is that there are parts that we can still modify. These are the very, I think, positive parts of patients’ recovery where we can still find places for optimization.
Puder:
I'm glad you asked. So let's say you had a provider who had borderline personality disorder. In general, someone with borderline personality disorder has lower reflective function. They start out, in this one study, on transference focused psychotherapy (TFP) [see episodes 234 and 239] versus dialectical behavioral therapy (DBT) versus supportive psychotherapy (Levy et al., 2006). They start out at around 2.8, people with borderline personality disorder. So this is a scale, remember from negative one to nine. So 2.8 is a low score. So by the end of the study, by one year of twice a week psychotherapy, they're able to go up to about a 4.1 with the transference-focused psychotherapy. So think about like a provider who is in years of therapy, years of good therapy, they will continually, hopefully increase their reflective function score and if they're utilizing good supervision and if they're approaching life with this kind of roadmap on how to increase their reflectiveness, then yes, I think absolutely, I think anyone can increase their reflectiveness. I think there are some people who probably start out higher than other people, obviously. I think that it doesn't seem to be related to IQ. People who go to prison have lower reflective function scores. People with alexithymia have lower reflective function scores. People with eating disorders have lower reflective function scores, and people with BPD have lower reflective function scores. So there are pathways of treatment for all of these types of groups of people that I just spoke about. And so getting effective treatment can, over time, increase your reflective function.
Puder:
So yes, I think it's hopeful. I think it's not as easy as you're going to go to a weekend seminar. You're going to learn some empathy skills because empathy breaks down, especially in close attachment relationships. Right? Like you could have a provider, for example, who's very empathic towards their patients, but maybe struggles with their significant other. Why are they struggling with their significant other? Because of their early attachment relationships? Maybe because that gap in reflective function with their early attachment relationships would cause them to struggle with later relationships as well in this, in a similar way. And so maybe they know how to empathize with people they have some distance with. Right? But when they get into a close interpersonal exchange with a patient, maybe a more sick patient, maybe that's when the countertransference comes up, that's when the attachment and machinery comes up that's unprocessed. So that's why there seems to be…. Actually, if you look at the research on therapist effect, it seems like there's a higher therapist effect with more sick patients than with mildly ill patients. As the illness goes up. Right?
de Beer:
Yes, exactly. And I think the same holds true for paranoia, as paranoia goes up. So in more severe cases, the therapist effect goes up.
Puder:
Yes, because it can be very jarring. It can be very confusing on what to do. Therapists are not often trained on how to deal with psychotic patients. I have a therapist that works for me now, who, when he first started working for me, he was like, “The patients that you're sending me are very ill compared to every patient I've seen in the past.” And it's because the patients that come to a psychiatrist, especially maybe like the ones that maybe I attract, especially ill ones, because of the podcast and because people who find me are really seeking for something, you know, that maybe they haven't gotten. So he says, “The patients that you're sending me are really ill.” And I've had to help him work with learning how to be patient with the psychotic symptoms and how to deal with them. And so there is a degree of specialized training and getting used to treating that population. I think that is very vital that providers need to go through. Let me ask you, how did you get interested in this though? Because it seems like you're more on the research side, but there's a fascination with this. Right? You're not treating, are you treating patients? Are you providing therapy for these clients, or are you mostly researching?
de Beer:
I'm a hundred percent research. Yes. Neuroscientist by training, but I feel very interested in how people perceive and experience the world. I can be studying the brain, of course, I'm studying the brain, but I am studying the brain to understand how and why people experience certain environments and ways the way they do. And now, I'm very interested also in improving treatments and lives of patients with psychosis. And I think it is, I think psychotherapy is such a personal and social experience, and I feel that if psychiatrists are just left out of the picture, while there is such a massive part of how a person will respond to the treatment, we are missing something completely.
de Beer:
I wanted to at least draw attention to this because I think it's an overlooked issue, surely. I think people should be more aware of how they are as a person, important in a patient's treatment, and how they, as a person, can also make a positive benefit. And for instance, the trainings you mentioned, that there is also room for improvement. So if you have this positive ability, maybe you have the complete positive potential, you can reach for or you can also strive to become even a better psychiatrist. And that does not only entail knowing what type of drugs and what type of doses to prescribe, that also entails knowing how you should respond, how you should deal with patients. And that interpersonal part is something that's so crucial and that may very well need lifelong learning rather than studying guidelines in the evening.
Puder:
Have you heard of Michael Garrett? He came on my podcast [see episode 180], great book on psychotherapy for psychosis, integrating CBT and psychodynamic [Psychotherapy for psychosis:
Integrating cognitive-behavioral and psychodynamic treatment (Garrett, 2019)]. Have you heard of him? He's a great resource. If anyone's listening to this and wants to go back to that episode to hear, some of his insights were really profound. And yeah, I'd like to do more. I think there is a certain level of just knowing about psychosis, knowing about for example, I'm doing an episode, it may air before yours, on delusions [see episode 261]. Two hours of Dr. Cummings, who works at a state hospital. So it's the most severe of severe patients. The patients who murdered someone or had some crime with a psychotic episode involved and led to being hospitalized extensively for years.
Puder:
So to look at the most ill of ill patients can sometimes give us some more patience, some more understanding, some more calm. Right? When we're treating patients. Because one of the things that we try to help families do with patients with schizophrenia is to be calm and to find their own peacefulness. And if they're yelling, if they're agitated, their son or daughter with schizophrenia is not going to do as well. Right? And so this is one of the things I think we could do as providers is to find our own ability to become very calm in the midst of someone else's psychotic ramblings, psychotic thoughts. Any further reflections or shall we say goodbye for today? It was very nice to meet you.
de Beer:
It was great talking to you.
Puder:
You're already churning out tons of great articles. I expect to have you back at some point as you publish more papers and you can continue to teach us and we can find an integration between the research and the brain science and how it can help us understand and help patients. So thank you for coming on.
de Beer:
Thank you for having me.
For further consideration:
Sommer, I. E., Gangadin, S. S., & de Beer, F. (2026). Interpretation of the HAMLETT study—Reply. JAMA Psychiatry, 83(3), 323–324. https://doi.org/10.1001/jamapsychiatry.2025.4450
References
Begemann, M. J. H., Thompson, I. A., Veling, W., Gangadin, S. S., Geraets, C. N. W., van ’t Hag, E., Müller-Kuperus, S. J., Oomen, P. P., Voppel, A. E., van der Gaag, M., Kikkert, M. J., van Os, J., Smit, H. F. E., Knegtering, R. H., Wiersma, S., Stouten, L. H., Gijsman, H. J., Wunderink, L., Staring, A. B. P., … Sommer, I. E. C. (2020). To continue or not to continue? Antipsychotic medication maintenance versus dose-reduction/discontinuation in first episode psychosis: HAMLETT, a pragmatic multicenter single-blind randomized controlled trial. Trials, 21(1), 147. https://doi.org/10.1186/s13063-019-3822-5
Chen, E. Y., Hui, C. L., Lam, M. M., Chiu, C. P., Law, C. W., Chung, D. W., Tso, S., Pang, E. P., Chan, K. T., Wong, Y. C., Mo, F. Y., Chan, K. P., Yao, T. J., Hung, S. F., & Honer, W. G. (2010). Maintenance treatment with quetiapine versus discontinuation after one year of treatment in patients with remitted first episode psychosis: randomised controlled trial. BMJ (Clinical research ed.), 341, c4024. https://doi.org/10.1136/bmj.c4024
Cologon, J., Schweitzer, R. D., King, R., & Nolte, T. (2017). Therapist reflective functioning, therapist attachment style and therapist effectiveness. Administration and Policy in Mental Health and Mental Health Services Research, 44(5), 614–625. https://doi.org/10.1007/s10488-017-0790-5
de Beer, F., Wijnen, B., Wouda, L., Koops, S., Gangadin, S., Veling, W., van Beveren, N., de Haan, L., Begemann, M. J. H., & Sommer, I. E. C., & HAMLETT-OPHELIA consortium. (2024a). Antipsychotic dopamine D2 affinity and negative symptoms in remitted first episode psychosis patients. Schizophrenia Research, 274, 299–306. https://doi.org/10.1016/j.schres.2024.09.030
de Beer, F., Koops, S., Schoevers, R. A., Veling, W., van Beveren, N., de Haan, L., Begemann, M. J. H., & Sommer, I. E. C. (2024b). Psychiatrists effect on positive symptom severity and daily functioning during pharmacotherapy for first-episode psychosis patients. Scientific Reports, 14(1), Article 22871. https://doi.org/10.1038/s41598-024-72678-4
de Beer, F., de Vries, E., Wijnen, B., Begemann, M. J. H., van Beveren, N., Boonstra, N., Gangadin, S. S., de Haan, L., Hamers, I. M. H., Veling, W., Koops, S., & Sommer, I. E. C. (2025). Dopamine D2/3R availability after discontinuation of antipsychotic treatment: A [11C]raclopride PET study in remitted first-episode psychosis patients. Psychological Medicine, 55, Article e264. https://doi.org/10.1017/S003329172510161X
de Beer, F., Hamers, I. M. H., Prycka, M., Schoretsanitis, G., Gangadin, S. S., Touw, D. J., & Sommer, I. E. C. (2025b). Trajectories of Clozapine Concentrations in Women Across Menopausal Age, Schizophrenia Bulletin. Advance online publication. https://doi.org/10.1093/schbul/sbaf186
de Beer, F., Brand, B. A., Wijnen, B., Gangadin, S. S., Schoretsanitis, G., Touw, D., Sommer, I. E. C., & PHAMOUS investigators (2026). The Impact of Sex and Age on Antipsychotic Serum Concentrations. Schizophrenia bulletin, 52(2), sbaf217. https://doi.org/10.1093/schbul/sbaf217
Gangadin, S. S., de Beer, F., Wijnen, B., Begemann, M., van Beveren, N., Boonstra, N., de Haan, L., Kikkert, M., Koops, S., van Os, J., Veling, W., & HAMLETT-OPHELIA Consortium. (2025). Risk of relapse during tapering of antipsychotic medication after a first psychotic episode: Association with D2 receptor affinity but not with tapering speed. World Psychiatry, 24(2), 240–249. https://doi.org/10.1002/wps.21315
Garrett, M. (2019). Psychotherapy for psychosis: Integrating cognitive-behavioral and psychodynamic treatment (1st ed.). Guilford Press. https://www.guilford.com/books/Psychotherapy-for-Psychosis/Michael-Garrett/9781462540563
Hui, C. L. M., Honer, W. G., Lee, E. H. M., Chang, W. C., Chan, S. K. W., Chen, E. S. M., Pang, E. P. F., Lui, S. S. Y., Chung, D. W. S., Yeung, W. S., Ng, R. M. K., Lo, W. T. L., Jones, P. B., Sham, P., & Chen, E. Y. H. (2018). Long-term effects of discontinuation from antipsychotic maintenance following first-episode schizophrenia and related disorders: A 10-year follow-up of a randomised, double-blind trial. The Lancet Psychiatry, 5(5), 432–442. https://doi.org/10.1016/S2215-0366(18)30090-7
Levy, K. N., Meehan, K. B., Kelly, K. M., Reynoso, J. S., Weber, M., Clarkin, J. F., & Kernberg, O. F. (2006). Change in attachment patterns and reflective function in a randomized control trial of transference‑focused psychotherapy for borderline personality disorder. Journal of Consulting and Clinical Psychology, 74(6), 1027–1040. https://doi.org/10.1037/0022-006X.74.6.1027
McKay, K. M., Imel, Z. E., & Wampold, B. E. (2006). Psychiatrist effects in the psychopharmacological treatment of depression. Journal of Affective Disorders, 92(2–3), 287–290. https://doi.org/10.1016/j.jad.2006.01.020
Sommer, I. E., de Beer, F., Gangadin, S., de Haan, L., Veling, W., van Beveren, N., Boonstra, N., Rosema, B.-S., van Os, J., Kikkert, M., Koops, S., Noorman, J., Thielen, F., Wijnen, B., & Begemann, M., for the HAMLETTT-OPHELIA Consortium. (2026). Early dose reduction or discontinuation vs maintenance antipsychotics after first psychotic episode remission: A randomized clinical trial. JAMA Psychiatry, 83(1), 68–73. https://doi.org/10.1001/jamapsychiatry.2025.2525
Sommer, I. E., Gangadin, S. S., & de Beer, F. (2026). Interpretation of the HAMLETT study—Reply. JAMA Psychiatry, 83(3), 323–324. https://doi.org/10.1001/jamapsychiatry.2025.4450
Taipale, H., Solmi, M., Correll, C. U., & Tiihonen, J. (2024). Relative Risk of Breast Cancer Associated With Prolactin-Increasing Antipsychotic Use. The Journal of clinical psychiatry, 85(1), 23lr15135. https://doi.org/10.4088/JCP.23lr15135
Wunderink, L., Nieboer, R. M., Wiersma, D., Sytema, S., & Nienhuis, F. J. (2013). Recovery in remitted first-episode psychosis at 7 years of follow-up of an early dose reduction/discontinuation or maintenance treatment strategy: Long-term follow-up of a 2-year randomized clinical trial. JAMA Psychiatry, 70(9), 913–920. https://doi.org/10.1001/jamapsychiatry.2013.19