Episode 246: Cannabis and Depression, Anxiety, PTSD, Sleep, and Cognitive Function Update and Review
Danielle Liu*, Daniel Cuevas**, Liam Browning, MD; Christopher Campbell, MD; David Puder, MD
Corresponding Author: David Puder, MD
Reviewers: Erica Vega, Joanie Burns, PMHNP-BC
*Danielle Liu is the first author on the sections on depression, anxiety, and PTSD
**Daniel Cuevas is the first author on the sections on cognitive function and sleep
The authors declare no conflicts of interest and have no financial relationships with any pharmaceutical or cannabis companies.
By listening to this episode, you can earn 1.25 Psychiatry CME Credits.
Other Places to listen: iTunes, Spotify
At a time when cannabis is not only legalized, but marketed as therapeutic, it is essential for healthcare professionals to be well-versed on what the research truly reveals about the reality of cannabis and its effect on psychiatric outcomes. Given the increased accessibility and its widespread popular appeal, it is incredibly common for patients to identify cannabis as one of the few things that alleviate their symptoms of anxiety and depression.
However, real-world scenarios highlight the true clinical effect that cannabis has on patients’ mental health journeys, and the importance of this issue. Take the case of the patient whose family members encourage him to continue using cannabis because without it, he becomes a “different person” - far more irritable and anxious and completely unable to function because of his overwhelming emotions. This is not a picture of a patient for whom cannabis is healing, but destructive.
Cases like this have been increasingly seen, raising relevant questions about the true effects of cannabis on depression, anxiety, PTSD, and other mental health outcomes. Can cannabis cause depression? Is cannabis effective for anxiety? How does cannabis affect sleep and PTSD symptoms? What are the best ways to identify and treat cannabis use disorder? In this episode, we continue our discussion on the psychiatric risks of cannabis (see episode 240) by examining the latest scientific research on cannabis’s impact on mood disorders, PTSD, sleep, and cannabis use disorder, and discuss important considerations for treating patients with problematic cannabis use.
Cannabis And Depression
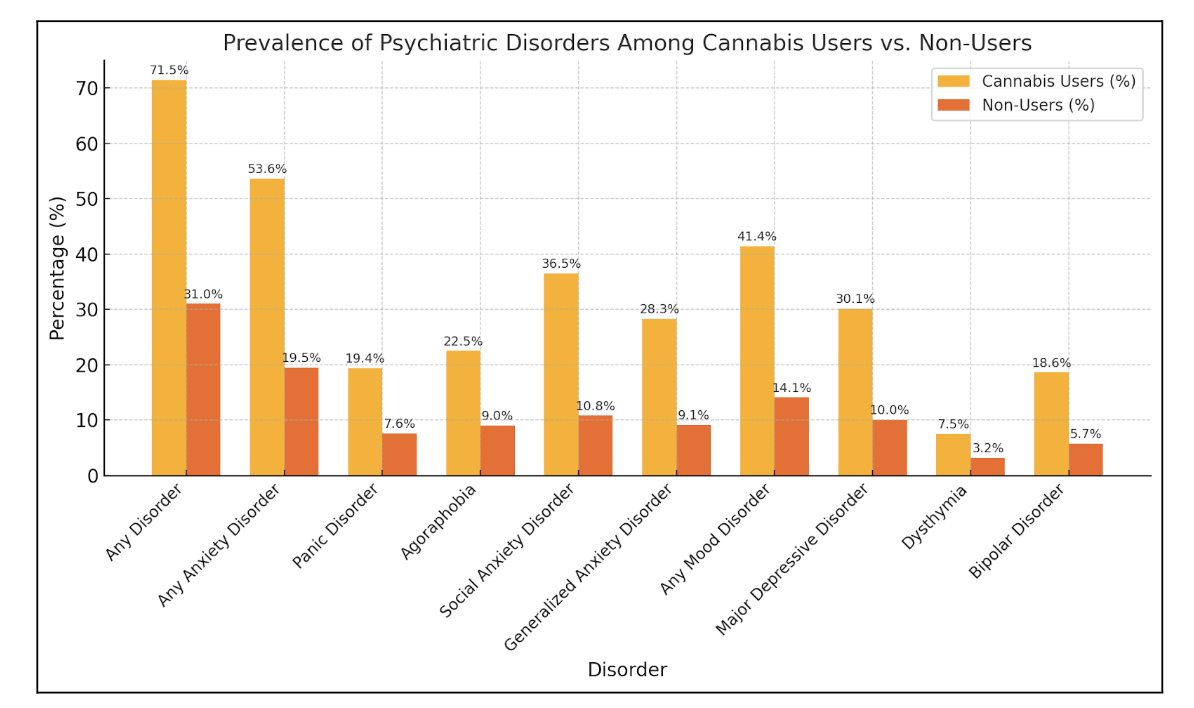
Cannabis use is correlated with increased likelihood of virtually all psychiatric disorders, as illustrated below in Figure 1 (Blanco et al., 2016). This is particularly concerning given the fact that marijuana use is associated with a myriad of deleterious mental health outcomes. When looking at the specific example of cannabis use in individuals with depression, it was found that over 30% of people with depression reported using cannabis in the past 30 days, and more than 15% reported daily use. Moreover, this study highlighted an alarming increase in cannabis use rates in individuals with depression in recent years: From 2005 to 2006, people with depression were 46% more likely to use cannabis and 37% more likely to use it nearly every day. By 2015 to 2016, these odds had increased significantly, with individuals with depression being 130% more likely to use cannabis and 216% more likely to use it daily (Gorfinkel et al., 2020).
Figure 1: Prevalence of psychiatric disorders among cannabis users compared to non-users. This figure highlights the percentage of individuals diagnosed with various psychiatric disorders within the past year, comparing cannabis users and non-users. Disorders include broad categories like "Any Disorder," "Any Anxiety Disorder," and "Any Mood Disorder," as well as specific subtypes such as panic disorder, social anxiety disorder, and bipolar disorder. Data adapted from Cannabis Use and Risk of Psychiatric Disorders Prospective Evidence From a US National Longitudinal Study (Blanco et al., 2016).
Let’s delve into what the research shows us about the relationship between cannabis use and depression.
Are depression and CUD associated?
People with Depression are more Likely to Have a Cannabis Use Disorder
A cross-sectional study done of the survey results from over 16,000 U.S. adults, conducted from 2005-2016 found that those with moderate-to-severe depression (per PHQ-9 scores) had nearly double the odds (OR 1.90 95% CI, [1.62-2.24]) of any past-month cannabis use and over triple the odds of daily/near-daily use (OR 3.16 95% CI [2.23-4.48]), compared to those without depression (Gorfinkel et al., 2020). Interestingly, this association grew stronger from 2005 to 2016, which may suggest that as public perception of cannabis as a “helpful” remedy increased, more depressed individuals turned to it.
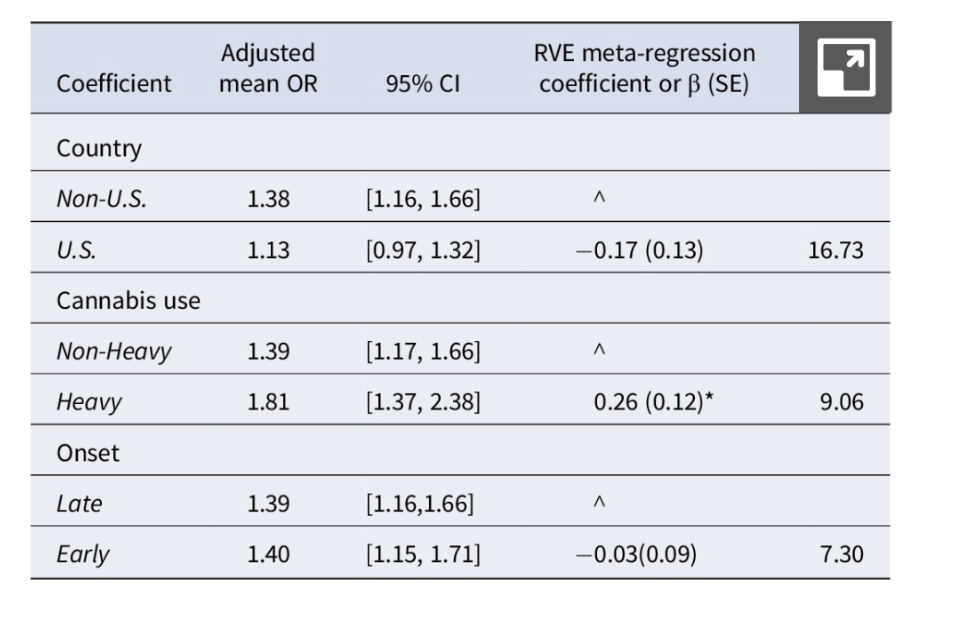
A meta-analysis published in February 2025 looked at the association between cannabis use (coding for usage prior to the age of 18 and heavy cannabis use, which was defined as “cannabis dependence,” “cannabis use disorder,” “diagnosis of cannabis abuse,” “chronic cannabis use,” “persistent use,” and “daily use”) in any form and self-reported or clinical depression in 22 studies. The analysis used multilevel meta-regression for effect size moderators. Cross-sectional studies and studies that focused on only special populations [including war veterans, LGBTQ-only populations, or participants seeking treatment for a baseline mental health issue] were excluded (to reduce confounding factors). This study showed that compared to people who do not use cannabis, people who use cannabis are more likely to have self-reported or clinical depression (OR 1.29, 95% CI 1.13-1.46) (Churchill et al., 2025).
Note. Reprinted from “The association between cannabis and depression: An updated systematic review and meta-analysis”, by Churchill et al., 2025, Psychological Medicine, 55, e44. https://doi.org/10.1017/S0033291724003143
The association between cannabis use and depressive symptoms was also corroborated by a systematic study from April 2024 which examined 78 studies. Although this study inherently was limited by the heterogeneity of the studies included and the cross-sectional design of a number of the included studies, the review still demonstrates the trend of cannabis use in people with mood disorders and suggests that cannabis use may be associated with poorer prognosis in major depressive disorder (Sorkhou et al., 2024). There is also a 2023 JAMA study that shows that CUD is associated with higher odds of depression (Jefsen et al., 2023).
A 2021 meta-analysis of epidemiological surveys representing the United States shows that the prevalence of comorbidity between cannabis use and major depression varies between recreational, medical and heavy cannabis use: the prevalence of comorbidity was 6.9% with cannabis dependence, with an odds ratio of 4.83 (95%CI 2.37 – 2.85); 4.7% with cannabis use disorder, (odds ratio of 2.6, 95%CI 2.37 – 2.85) and only 1% with cannabis abuse (odds ratio 2.37, 95% CI 1.38 – 4.07) (Note: Although cannabis and abuse are now under the same category under the updated DSM V, some of the studies included in this meta-analysis were published before CUD was defined) (Onaemo et al., 2021).
However, these studies do not tell us whether cannabis causes depression. RCTs are limited in cannabis research, so we are stuck with mostly observational studies. To understand the relationship between cannabis and depression, we can look at studies meeting the Bradford Hill criteria of dose-response (higher potency, greater frequency of use) and temporality (cannabis use preceding onset of depression) along with controlling for confounds (socioeconomic, adverse childhood experiences, genetic factors).
If someone starts smoking cannabis, does it increase their risk of developing depression?
Twin Studies
Twin studies assessing two different monozygotic twins who are discordant for cannabis use offer unique ways of controlling for genetic and environmental confounds because monozygotic twins share the same genetics and have a more similar environment than other controls.
A large retrospective study of 6181 monozygotic and dizygotic twins showed a greater frequency of reported MDD in twins who used cannabis more frequently (>100 times vs. <100 times), with an odds ratio of 1.98 (95% CI [1.11–3.53]) (Agrawal et a.l, 2017). However, when looking at only the 198 discordant monozygotic twins and adjusting for covariates like conduct disorder, early dysphoria or anhedonia, sexual abuse, and alcohol use, the odds ratio fell to 1.68 (95% CI [1.01-2.80]).Given that the confidence interval reflected marginal significance, this may represent a case of p-hacking, given that such an arbitrary number of 100 uses of cannabis was used to result in significance in this study.
The Minnesota Twin Study by Schaefer et al., (2021) examined adolescent cannabis use and its long-term psychosocial impact in a sample of over 3,700 twins.
This study found that when comparing monozygotic twins, differences in cannabis use were significantly associated with lower educational attainment, mainly through decreased grade point average, decreased academic motivation, increased academic problem behavior, and increased school disciplinary problems. Furthermore, these same outcomes were associated with poorer socioeconomic outcomes later in young adulthood.
Interestingly, this study found that adverse psychiatric outcomes (such as MDD, anxiety disorder, antisocial personality disorder, other substance use disorder), as well as poorer performance on a vocabulary task, were each associated with increased cannabis use; however, these associations did not persist when comparing monozygotic twins on their cannabis use.
One limitation here is that for the MZ co-twin analyses only a small amount of cognitive tests were performed, and thus may not accurately represent a full cognitive assessment.
Important as IQ would be more encompassing and is one of the strongest individual predictors of career performance, academic achievement, and socioeconomic status.
Take-home: Adolescent cannabis use appears to causally undermine academic functioning and later socioeconomic success, but evidence from twin designs indicates that its associations with depression, anxiety, (and perhaps baseline cognitive ability), more-so reflects underlying familial and genetic predispositions rather than a direct neurotoxic effect.
Effects of Cannabis Use in Adolescents and Prospective Studies
A 2019 meta-analysis looking specifically at prospective longitudinal studies from inception to January 2017 investigated the association between cannabis use in adolescence (<18) and risk of depression, anxiety, and suicidality. The study found that cannabis use in adolescence is associated with increased risk for depressive disorder in young adulthood, the odds ratio being 1.37 for users compared to non-users (95% CI 1.16-1.62) (Gobbi et al., 2019). However, this study is limited by its high heterogeneity. Further, many of the included studies did not adjust for other drugs of abuse and cigarettes, genetic predisposition, personality disorder, or psychosocial factors (peer drug abuse, school abandonment) which can independently contribute to major depressive disorder.
A previous meta-analysis of 14 prospective longitudinal studies controlled for many of these confounds (psychiatric symptoms, socioeconomic status) and also met the Bradford Hill criteria of dose-response and temporality, finding a smaller OR of 1.17 (95% CI 1.05–1.30 for developing depression in users vs. controls; but a greater OR of 1.62 (95% CI 1.21–2.16) in heavy cannabis users (at least weekly use) compared to non-users/light users (Lev-Ran et al., 2014).
How could cannabis make depression worse?
Another perspective to consider in the relationship between cannabis use and depression is withdrawal-related dysphoria, which can be highly disabling for dependent cannabis users and frequently leads to relapse (in order to alleviate the discomfort of withdrawals). Consequently, once a person is an established, heavy user of cannabis, he might feel down when he is not using, which then “requires” them to use again, reinforcing a cycle where a person needs to keep using cannabis just to feel “okay” and avoid feeling depressed. Is cannabis causing depression, or are people using cannabis to avoid feeling depressed due to withdrawal?
Suicide And Suicidal Ideation And Cannabis Use
A large retrospective study of 6181 monozygotic twins showed that suicidality in the monozygotic twin who used cannabis more frequently was more likely to be reported (OR 2.47, 95% CI 1.19–5.10) (Agrawal et al., 2017).
An integrative analysis of multiple longitudinal studies, comprising between 2500-3700 participants who had been serially assessed between ages 13-30, observed that participants who used cannabis before age 17 daily compared to participants who had never used cannabis had a dose-dependent increase in odds of suicide attempt by age 25-30 (OR: 6.83, CI 2.04-22.90). However, results were confounded by lower educational attainment, more illicit drug use, welfare dependence, depression, etc., so it is difficult to ascertain that cannabis was an dependent predictor of suicide attempt (Silins et al., 2014).
A 2019 meta-analysis looking at cannabis use in adolescence and the risk of suicidality found an increased risk of suicidal ideation in users versus non-users (with OR = 1.5, 95% CI 1.11-2.03). The most significant result was that the odds ratio for increased risk of suicide attempts in cannabis users vs. non-users was 3.46 (95% CI 1.53-7.84) (Gobbi et al., 2019). However, this study is limited by its high heterogeneity and an outlier study.
A 2017 population-based longitudinal study found a significant difference in thoughts about committing suicide (for non-cannabis users vs. cannabis users without CUD, χ2= 7.54, p=0.0078; for non-cannabis users vs. participants with CUD, χ2= 7.44, p=0.0082), but after adjusting for more baseline confounding factors, this difference did not retain significance, implying that much of the seemingly apparent association between cannabis use and MDD may in fact be associated with sociodemographic and clinical factors instead of cannabis use (Feingold et al., 2017).
A longitudinal sample of 6788 adolescents, which followed participants from ages 14-15 to 16-17 and looked at their NLSCY data, found that at least monthly cannabis use at age 14-15 was associated with increased SI (OR 1.74 [1.16, 2.60]) and suicide attempts (OR 1.87 [1.09, 3.22]) two years later in adolescents. In the study, participants had no recent history of depression, and the study controlled for other drug use, life stress, maternal depression, and family dysfunction. This suggests cannabis use’s effect on suicidal ideation may actually be independent of depressive symptoms (Weeks & Colman, 2017).
Another study which followed 3134 adolescents aged 11-17 for one year identified marijuana use within the past year (OR = 4.7) and caregiver suicide attempts as the only independent predictors of the first incidence of suicide attempts (Roberts et al., 2010). The major limitation of this study is that no dose-response relationships were investigated.
A longitudinal study following New Zealanders born in 1972 and 1973 originally looked at the association between multiple variables and suicidal ideation, focusing on tobacco usage. In this study, only depressed mood, high stress, and low parental attachment at age 15 were significant predictors of suicidal ideation (McGee et al., 2005); cannabis use was not detailed, but this points to how a number of confounders also are independently associated with suicidal ideation as well, which were not accounted for in other studies.
Anxiety And Cannabis Use
A meta-analysis of 24 longitudinal and prospective studies assessing adolescent cannabis use and later anxiety disorder found increased odds of developing anxiety on follow-up (OR = 1.25; 95% CI, 1.01 to 1.54; I2 = 39%), with a reduction in this OR when the analysis was restricted to studies that used a structured diagnostic interview instead of self-reported questionnaires (OR = 1.15; 95% CI, 1.01 to 1.30) (Xue et al., 2021). Restricting the analysis to high-quality studies, or to studies which considered cannabis use prior to age 18, or to studies published after 1999, resulted in a loss of statistical significance. In general, the ORs are not very impressive, suggesting that there is at best weak evidence that cannabis increases risk for anxiety disorders. Given that the association did not retain significance when cannabis use prior to age 18 was accounted for, it is unlikely that early cannabis use is a significant causative factor in the development of anxiety disorders later in life.
If someone starts smoking cannabis, does it increase their risk of developing anxiety disorders?
Another 2020 literature review of prospective studies acknowledges a robust positive association between cannabis use and anxiety symptoms and disorders, but points out that minimal research has been suggestive of any causative or predictive relationship in either direction, noting that any evidence supporting a directional relationship (for the most part) does not (for the most part) appropriately adjust for covariates and confounding factors (Garey et al., 2020). One prospective epidemiological analysis of past-year weekly cannabis use did predict panic disorder with agoraphobia (but not without) after adjusting for comorbid psychiatric disorders and sociodemographic factors (OR 1.56, CI 1.11-2.19) (Cougle et al., 2015), and another study noted that daily or almost daily cannabis use marginally predicted the onset of SAD (OR 4.62, CI 2.29-9.31 in unadjusted analysis; OR 2.88, CI 1.4-5.77 after adjusting for sociodemographic variables; OR 2.24, CI 1.12-4.48 after additionally adjusting for alcohol and substance use disorders; OR 1.06, CI 0.99-3.94 after additionally adjusting for comorbid psychiatric disorders) (Feingold et al., 2016), but aside from these studies, such predictive relationships with robust studies have been sparse in the literature.
Directional relationship between anxiety and cannabis use
A more recent longitudinal cohort study published in 2022 demonstrated that in the overall study population and in men, increased cannabis use predicted greater increases in anxiety at a later time point. In contrast, more severe anxiety symptoms were associated with decreasing cannabis use. In females, when prior levels of trait-like anxiety predicted increased anxiety symptoms, cannabis use was found to decrease among women who recently experienced worsening anxiety (Davis et al., 2022).
Cannabis To Treat Depression And Anxiety
Cannabis for Depression
On the flip side, some have argued that cannabis can be used to actually treat depression. One observational study used data from Strainprint, a medical cannabis app that users input their depression, anxiety, and stress symptoms and tracked their symptoms over time. Twenty minutes after cannabis use, the patients reported alleviation in their symptoms of depression, but longer-term improvements were not found, emphasizing the argument against using cannabis to treat depression (Cuttler et al., 2018).
A single-blind randomized clinical trial study in Massachusetts found that getting a medical marijuana card led to increased cannabis use and higher rates of cannabis use disorder symptoms, without significant improvement in pain or depression after 12 weeks. Depression was measured via the Hospital Anxiety and Depression Scale (21 points total), and the study compared scores for people who immediately received access to a medical marijuana card versus those who received it 12 weeks later. Scores were recorded at the beginning of the study, week 2, week 4, and week 12, and the mean difference in the depression score was only -0.5 (p=0.3) (Gilman et al, 2022).
Because cannabis can cause patients to feel that their symptoms are improving initially without long-term benefit, using cannabis may in fact play a role in preventing people from getting better.
Cannabis for Anxiety
Anxiety is one of the most common reasons cited by medical marijuana patients for their cannabis use. The study, which used the medical cannabis app to track usage and symptom response, found a significant reduction in the ratings of anxiety (MBefore = 5.98, SE = 0.12 vs. MAfter = 2.50, SE = 0.14), Wald χ2 (1, 769) = 659.50, p < .001), with reported reduction in anxiety in 93.5% of tracked sessions. Anxiety was exacerbated in 2.1% of sessions, and there was no change in anxiety symptoms for 4.4% of sessions. This difference was observed in both men (Wald χ2 (1, 407) = 244.61 and women (Wald χ2 (1, 362) = 582.32, p < .001), with women perceiving a greater reduction in anxiety (Wald χ2 (1, 362) = 10.78, p < .001) (Cuttler et al., 2018).
The randomized clinical trial in Massachusetts found no significant difference in anxiety symptoms after treatment with medical marijuana after 12 weeks. Anxiety was measured via the Hospital Anxiety and Depression Scale like depression was, and the mean difference for anxiety was only -0.1, (p=0.9). Of course, there was individual variability, and some participants found benefit from the drug, but overall, there was no significant improvement found. Further research is needed to be done in order to direct future medical uses of marijuana (Gilman et al., 2022).
PTSD And Cannabis Use
Although medical marijuana is approved to treat PTSD in some states, the VA/DoD Clinical Practice Guideline for PTSD (2023) recommended against the use of cannabis for the treatment of PTSD due to lack of evidence for its efficacy. Nevertheless, many patients report that cannabis helps with their symptoms or improves their overall life and functioning. We have heard patients state:
It helps them sleep because it reduces nightmares.
It has helped them avoid their previous addictions, such as alcohol or opioids.
It helps them look at themselves from a different perspective, not as negative and more in a positive light.
When they come off of it their symptoms are worse, they have more nightmares and are more “on edge.”
An excellent article on the U.S. Department of Veteran Affairs website written by Melanie Hill and colleagues in 2024(a) discusses the use of cannabis in patients with PTSD. They report that cannabis has not been found to improve PTSD symptoms, stating that in “a recent systematic review of 14 studies evaluating the evidence on the clinical effects of cannabis on PTSD symptoms, results did not support the use of cannabis for improving overall PTSD symptoms [(Rodas et al., 2024)].”
However, “there has only been 1 RCT comparing whole plant cannabis and placebo for treating PTSD [(Bonn-Miller et al., 2021)]”:
“This trial included 2 phases. The first phase compared effects of 3 active cannabis preparations (high THC, high CBD, balanced THC+CBD) and placebo [lowTHC/low CBD marijuana] on PTSD symptoms in 80 U.S. military veterans.”
Participants were allowed to self-administer ad libitum up to 1.8g, and there were no differences in grams used throughout the study. Although this was a double blind study, there was a 60% break-blind rate in the placebo group, 58% in the CBD group, and 100% in the high THC group.
After 3 weeks, all treatment groups showed improvement in PTSD symptoms and that there was “no significant difference in PTSD symptom reduction [CAPS-5] between placebo and any of the active cannabis preparations.”
All treatment arms saw a reduction in CAPS-5 scores:
High THC 15.2 points (SD = 11.3, p < .0001, d = -1.99)
Placebo 13.1 points (SD = 12.10, p < .001, d = -1.30)
THC+CBD 8.5 points (SD = 9.88, p < .05, d = -.83)
High CBD 8.4 points (SD = 10.09, p < .05, d = -.79)
After a two-week washout period, “74 veterans were re-randomized to receive 1 of 3 active cannabis preparations [High THC, High CBD, and THC+CBD]. Results showed a significant reduction in PTSD symptoms [PCL-5] in the THC+CBD group only; however, because there was no placebo group in this phase, it is not possible to draw conclusions about the efficacy of cannabis to treat PTSD from these results.”
It may be the case that cannabis reduces PTSD symptoms while the patient is intoxicated, as they cite a study in which 404 medical cannabis users with self-reported PTSD tracked their PTSD symptoms with an app and experienced “short-term symptom relief when using cannabis but no long-term changes in PTSD symptoms” over 31 months (LaFrance et al., 2020).
This raises the question of whether cannabis use impacts the effectiveness of therapy:
Evidence suggests that a diagnosis of CUD predicts less symptom change during residential PTSD treatment (Bonn-Miller et al., 2013) and continuing or starting to use cannabis after PTSD treatment has been linked to increased PTSD symptoms (Wilkinson et al., 2015). However, other studies have found no association between baseline cannabis use and post-treatment PTSD symptoms (Bedard-Gilligan et al., 2018; Ruglass et al., 2017). In a meta-analysis of 4 RCTs comparing trauma-focused and non-trauma-focused treatments for co-occurring PTSD and SUD, both cannabis users and non-users experienced clinically significant improvements in PTSD symptoms. Additionally, trauma-focused treatments were more effective than non-trauma-focused treatments, regardless of whether individuals used cannabis (Hill et al., 2024b). It is possible that functional problems related to cannabis use, rather than a neurobiological effect of cannabis, might impact PTSD treatment effectiveness. Individuals using cannabis may also have more difficulties engaging in treatment; one study found that baseline cannabis use predicted a doubled risk of dropout from both cognitive-behavioral and pharmacological treatments for PTSD, as well as poor adherence to trauma-focused psychotherapy (Bedard-Gilligan et al., 2018).
From these data, the authors conclude that “individuals with comorbid PTSD and SUD do not need to wait for a period of abstinence before addressing their PTSD. A growing number of studies demonstrate that these patients can tolerate trauma-focused treatment and that these treatments do not worsen substance use outcomes.”
Cannabis And Sleep
Patients commonly report subjective improvements in sleep with cannabis, and individuals with PTSD are especially likely to use it in an effort to reduce nightmares. Observational evidence suggests that cannabis can shorten sleep latency, increase slow-wave sleep, and decrease REM latency and duration. This suppression of REM may explain why vivid dreams are a hallmark of cannabis withdrawal.
However, these effects may not persist due to tolerance. Over time, the impact of cannabis on sleep often becomes neutral, and in some cases may worsen sleep quality. Evidence in this area is still limited and inconsistent.
To date, only 10 randomized controlled trials (RCTs) have directly examined cannabis and sleep outcomes (Amaral et al., 2023). These trials, which investigated different formulations of THC and CBD but not whole-plant cannabis, were short in duration (≤2 weeks) and had small sample sizes. Their findings were generally neutral to slightly positive.
A broader scoping review of 50 studies published up to 2023 (Amaral et al., 2023) found highly variable results: 21% reported improved sleep, 48% reported worse sleep, 14% reported mixed outcomes, and 17% found no effect. Taken together, the evidence suggests cannabis may provide short-term, subjective improvements, but tolerance, withdrawal effects, and inconsistent findings across studies highlight the need for further research.
Can Adding CBD or Terpenes to Cannabis Decrease Anxiety from THC? The “Entourage Effect”
CBD supplementation is common for people seeking ways to reduce symptoms of anxiety. Despite its popularity, there have only been a handful of small studies. One double-blind, placebo-controlled study evaluated the effect of the administration of 300 mg CBD daily for 4 weeks to 17 Japanese teenagers on social anxiety, using the scores of the Fear of Negative Evaluation questionnaire and the Liebowitz Social Anxiety Scale as outcome measures. The mean FNE score decreased from 24.4 to 19.1 after the CBD administration, compared to a decrease of 23.5 to 23.3 in the placebo group, with 2x2 ANOVA resulting in an F of 10.35 (p = 0.003) in the intervention group compared to the placebo group (F = 2.69, p=0.11) (Masataka, 2019). This suggests that CBD may have anxiolytic effects, although the power of this study was weak. Another randomized placebo-controlled study assessing the effect of 300 mg of CBD on anxiety (assessed via Visual Analog Mood Scale score) in participants with PTSD, as they listened to digital audio playback of their report of the triggering event, found that CBD significantly lessened the anxiety among the nonsexual trauma group (mean difference of 8.21, p = 0.008, CI -13.94 to -2.47), although this relationship was not found for sexual trauma (Bolsoni et al., 2022).
However, as concluded in a literature review exploring CBD and anxiety, evidence is mixed and limited (Lichenstein, 2022).
Notably, there are some ideas about CBD or terpenes resulting in different subjective effects (for example, altering the euphoric state or reducing anxiety) when compared to THC in isolation. The entourage effect theory posits that the non-THC components within whole-flower cannabis influence the overall effects of cannabis. Some believe that other phytochemicals and plant compounds may work synergistically to result in various effects which are different from that of any single compound.
Initial studies showed CBD could offset some of the negative intoxicating effects of high dose THC, like anxiety/paranoia:
Karniol and colleagues (1974) examined the effects of oral CBD by itself and on THC-induced effects. Although oral CBD (15, 30, and 60 mg) did not reduce anxiety on its own, it did reduce oral THC (30 mg) induced anxiety when given simultaneously. This was replicated by Zuardi and colleagues (1982) using a combination of oral CBD (1.0 mg/kg) and oral THC (0.5 mg/kg).
Solowij et al., 2019 (vaporized THC+ vaporized CBD), low-dose CBD potentiated the intoxicating effects of THC, high-dose CBD decreased intoxication, no effect on acute anxiety or psychosis.
However, more recent trials observed no effect on acute anxiety:
Hindocha and colleagues (2015) vaporized CBD+THC, CBD showed no effect on intoxication or anxiety.
Karschner and colleagues (2011) reported no effect of oral CBD (5-15mg) on oral THC-induced anxiety.
Arkell et al., 2021 (vaporized THC alone (13 mg) vs. CBD + THC [13 mg/13mg] vs. placebo). No difference in acute anxiety, subjective drug effects, cognitive performance.
And a recent trial published in JAMA (Zamarripa et al., 2024) suggested oral THC + high-dose oral CBD (20 mg THC + 640 mg CBD) increased plasma THC and hydroxy-THC concentrations, increased paranoia, and anxiety relative to THC alone (20mg).
However, all participants in this study were also taking a CYP-inhibiting cocktail, as the original intention of the study was to observe how these drugs interact with CBD and THC metabolism, not to answer the question of how CBD affects THC.
Nevertheless, oral THC relies more on first-pass metabolism than smoking/vaped THC, and produces more hydroxy-THC (active metabolite that’s stronger than THC). So, inhibiting the breakdown of THC and hydroxy-THC with a CYP-inhibiting cocktail and then adding CBD on top of that is likely going to prolong the availability of the psychoactive metabolite.
Of note, orally-dosed CBD has very poor bioavailability (Millar et al., 2018), and clinical doses of pharmaceutical grade CBD given for seizure disorders are 1000’s of mg (von Wrede et al., 2021), which dwarfs the amount used in supplements (5-20mg). For CBD to exert a biological effect, it may need to be taken at high doses, inhaled via smoke or vape, or taken as full-spectrum oil. Furthermore, the safety of vaping CBD-oil is in question, as it may produce toxic byproducts (Bhat et al., 2023; Love et al., 2024).
What are Terpenes?
Terpenes are a class of aromatic phytochemicals that give the plant its aroma and flavor. For example, limonene is found in lemons and citrus fruits, pinene is in pine needles, and myrcene is in hops. Terpenes are increasingly being artificially added to cannabis, with companies advertising them to lead to different “medicinal” effects, claiming some combinations of terpenes may be more calming while others may give an energized or focused effect, and that users should experiment with different terpene ratios and find what works for them. For instance:
This is questionable, as expectancy effects play a large role in how someone feels when they use a drug. We were only able to find one RCT addressing this:
A 2024 double-blind within-subject crossover study of 38 participants (10 males and 10 females of whom completed the study and were included in the data analysis) evaluated the effect of vaporized and inhaled THC alone (15 or 30 mg), d-limonene alone (1 or 5 mg), these compounds together, or placebo on the anxiogenic effects of THC in cannabis. The protocol also implemented an optional 10th test of 30 mg THC combined with 15 mg of d-limonene. The most promising results were observed with the 30 mg THC and 15 mg d-limonene combination, with significantly lower subjective ratings of the “paranoid” and “anxious / nervous” Drug Effect Questionnaire items compared to 30 mg THC alone (p’s < 0.05) (Spindle et al., 2024). This suggests that D-limonene may play a role in attenuating THC’s anxiogenic effects. In vitro binding studies have suggested that the attenuative effects of d-limonene do not directly modulate the effect of THC on the CB1 or CB2 receptors, but rather may act independent from CB receptors.
Cannabis, Cognition, And IQ
Another important consideration with regards to cannabis use in adolescents and young adults is its effect on cognition and intelligence.
A review and meta-analysis of the literature (69 studies) on cannabis usage and cognition in this demographic (Scott et al., 2018) came to the conclusion that THC can have statistically significant but clinically negligible effects on cognitive functioning for those who used cannabis frequently. However, these deficits seem to diminish given that periods of abstinence longer than 72 hours led to a decrease in effect as well as a loss in statistical significance. Thus, those who used cannabis but were abstinent had outcomes that were not measurably different from those who didn’t use cannabis.
Just like with the literature on cannabis’ effect on other aspects of mental health, there is not a consensus on the exact effects of cannabis on cognition and intelligence.
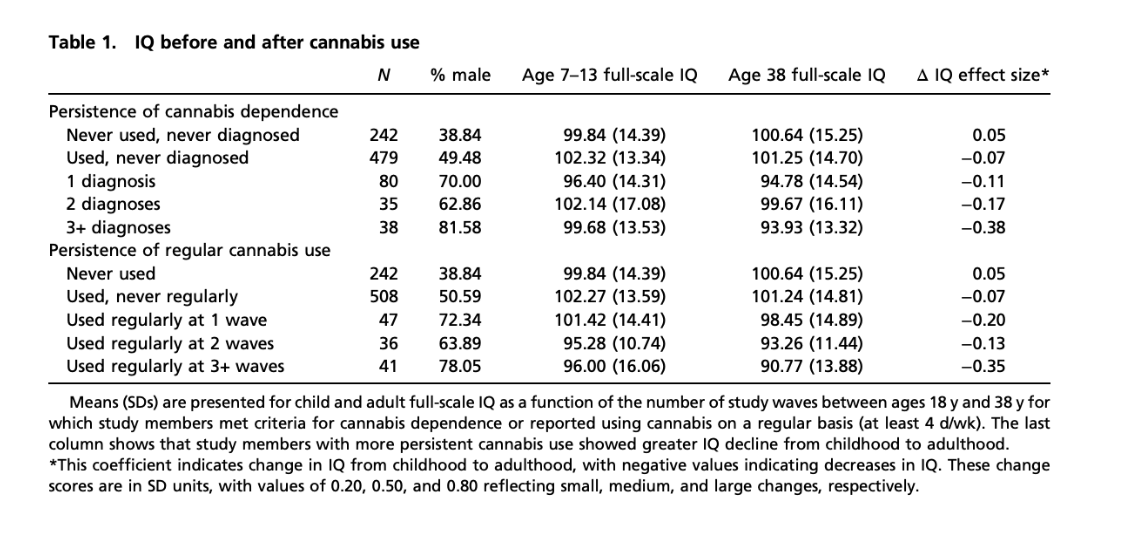
There was a prospective cohort study with 1,037 members of the Dunedin Cohort followed from birth (1972/1973) to the age 38 (Meier et al., 2012) that sought to test the association between persistent cannabis use and neuropsychological decline, as well as to determine whether decline is concentrated among cannabis users who started using in adolescence. Participants were evaluated regarding cannabis use through interviews at ages 18, 21, 26, 32, and 38 to assess for past-year cannabis dependence based on the criteria from the Diagnostic and Statistical Manual of Mental Disorders (DSM). Due to the fact that there were some members within the study who used cannabis on a regular basis but never met full criteria for a diagnosis of cannabis dependence, the researchers repeated analyses using persistent regular cannabis use as the exposure variable. Persistence of cannabis dependence was defined as the total number of study waves, out of five, at which a study member met criteria for cannabis dependence. Persistent cannabis use was associated with decline broadly across domains of neuropsychological functioning, even when controlling for years of education.
They found that impairment was particularly concentrated among adolescent-onset cannabis users, and more persistent use was associated with greater decline. Those who used cannabis persistently showed greater decline in IQ, and this is reflected in the fact that those who were diagnosed with cannabis dependence at one, two, or three or more waves within the study experienced IQ declines of −0.11, −0.17, and −0.38 SD units, respectively; for context, an IQ decline of −0.38 SD units corresponds to a loss of ∼6 IQ points. IQ decline was particularly pronounced in those who initiated cannabis use during adolescence, with adolescent-onset persistent cannabis usage (3+ diagnoses) was about −0.55 SD (~8 IQ points), which is significantly greater compared to adult-onset cannabis users (p = 0.02). Adolescent-onset persistent cannabis users showed significant IQ decline whether they used cannabis infrequently or frequently at age 38 (decline observed regardless of cessation, p = 0.03 for infrequent, p = 0.0002 for frequent use), whereas adult-onset persistent cannabis users did not show significant IQ decline with cessation (p = 0.73 for infrequent users, p = 0.11 for frequent users), suggesting possible recovery or less vulnerability.
Note. Reprinted from “Persistent cannabis users show neuropsychological decline from childhood to midlife”, by Meier et al., 2012, Proceedings of the National Academy of Sciences of the United States of America, 109(40), E2657–E2664.
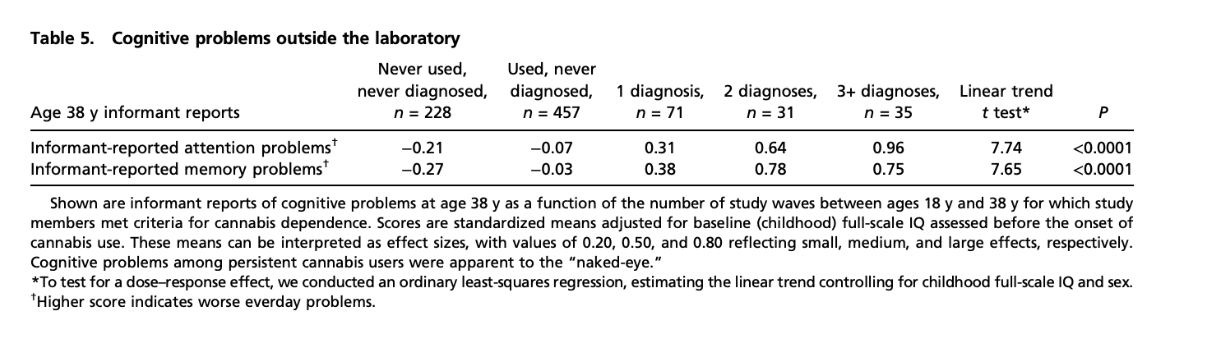
Additionally, study members with more persistent cannabis dependence generally showed greater neuropsychological impairment across different areas of mental function. For example, informants reported observing more attention and memory problems among those with more persistent cannabis dependence to a statistically significant degree. Furthermore, the linear effect of persistent cannabis dependence on change in full-scale IQ was significant before controlling for years of education (t = −4.45, P < 0.0001) and after controlling for years of education (t = −3.41, P = 0.0007). IQ decline could not be explained by other factors, even when controlling for hard-drug or alcohol dependence.
Note. Reprinted from “Persistent cannabis users show neuropsychological decline from childhood to midlife”, by Meier et al., 2012, Proceedings of the National Academy of Sciences of the United States of America, 109(40), E2657–E2664.
Note. Reprinted from “Persistent cannabis users show neuropsychological decline from childhood to midlife”, by Meier et al., 2012, Proceedings of the National Academy of Sciences of the United States of America, 109(40), E2657–E2664.
These findings are consistent with the hypothesis that cannabis use may have neurotoxic effects, especially in adolescence when the brain is undergoing crucial development. A limitation to this study was that it could not assess the concentration of THC being used. With regards to the Bradford Hill criteria, there seems to be a dose-response relationship in that more persistent use was associated with greater decline.
Notably, the ABCD study, an ongoing prospective study following 11,875 youth from ages 9-10 onward, also assessed cognitive function in a subsample (∼1,200 participants). A recent study of this subsample (Wade et al., 2024) found that compared to controls matched on socioeconomic status (parental education and household income), those with self-reported cannabis use or with positive metabolites in hair had slightly poorer episodic memory assessed via the Picture Memory task (F(1,238) = 4.67, partial η2 = 0.02, p = .03) compared to controls. Several years prior, these groups did not differ on their task performance. There were no significant differences in performance on other tasks related to verbal memory, working memory, attention inhibition, speed, and reading.
Cannabis Addiction: Cannabis Use Disorder, Withdrawal, And Treatment
How Addictive is Weed?
There is a popular belief that cannabis is not addictive; however, the evidence demonstrates this is not the case, as 8-19% of individuals who use cannabis have CUD (Hasin, 2018).
For comparison, about 15% of alcohol users develop an AUD (Grant et al., 2015).
And although more people use alcohol and have a use disorder with alcohol, there are now more people who use cannabis daily, showing that it may be particularly rewarding for a subpopulation of cannabis users.
THC’s rewarding effects are attributable to a reduction of GABAergic inhibition of VTA dopaminergic neurons (disinhibition).
Some studies have even shown there are genetic predispositions that cause people with schizophrenia and their healthy relatives to release more dopamine and glutamate in the striatum in response to THC (Colizzi et al., 2020; Kuepper et al., 2013).
Cannabis Use Disorder Diagnosis, Treatments And Withdrawal
Assessing a Patient for Cannabis Use Disorder
When assessing a patient for CUD, clinicians can use the DSM–5, which defines cannabis use disorder as the presence of clinically significant impairment or distress for 12 months, manifested by at least two of 11 criteria for a diagnosis of mild CUD, 4-5 symptoms for moderate CUD, and 6+ for severe CUD (Patel & Marwaha, 2024):
Taking cannabis in larger amounts or over a longer period than intended.
Persistent desire or unsuccessful efforts to cut down or control cannabis use.
Spending a great deal of time obtaining, using, or recovering from cannabis.
Craving or a strong desire to use cannabis.
Recurrent cannabis use resulting in failure to fulfill major role obligations at work, school, or home.
Continued cannabis use despite having persistent or recurrent social or interpersonal problems caused or exacerbated by cannabis.
Giving up or reducing important social, occupational, or recreational activities because of cannabis use.
Recurrent cannabis use in situations where it is physically hazardous.
Continued cannabis use despite knowledge of having a persistent or recurrent physical or psychological problem likely caused or exacerbated by cannabis.
Tolerance, as defined by either:
A need for markedly increased amounts of cannabis to achieve intoxication or desired effect.
A markedly diminished effect with continued use of the same amount of cannabis.
Withdrawal, as manifested by either:
The characteristic withdrawal syndrome for cannabis (e.g., irritability, anxiety, sleep difficulties, decreased appetite, restlessness, depressed mood, or physical symptoms such as abdominal pain or tremors).
Cannabis (or a closely related substance) is taken to relieve or avoid withdrawal symptoms.
As a clinician, when collecting a history on patients who use cannabis, it is important to assess the following issues during the clinical interview:
Onset of Use: When did the patient first try or regularly start using cannabis?
Frequency and Quantity: How often do they use? How much in a typical session (e.g., number of joints, grams, vape cartridges)?
Route of Administration: Smoking, vaping (flour vs. oil), edibles, highly concentrated forms of THC (dabs)
Patterns and Context: Where and with whom do they use? Are there certain triggers (stress, social pressure, boredom)? What do they find helpful about using?
Previous Treatment Attempts: Have they tried to quit or reduce use before? What worked, what didn’t?
Functional Impact: Are they missing work or classes due to their use? Are they having more conflicts with friends and family? (this is also particularly important, because by delineating the negative consequences associated with their use you can help cultivate motivation for behavioral change)
What are the symptoms of cannabis withdrawal?
Prevalence estimates on the proportion of patients with CUD who develop withdrawal symptoms after cessation vary from 8-100% depending on the study (Bahij et al., 2020), but these withdrawal symptoms are typically observed only in those with daily or near daily use.
Cannabis withdrawal symptoms typically occur within the first 24-48 hours after cessation, peak at days 2-6, and can continue for up to three weeks. Fortunately, cannabis withdrawal tends to be mild and is not often associated with significant adverse outcomes (Connor et al., 2022), and the typical symptoms can include disruptions in sleep (such as insomnia, vivid dreams), mood changes (such as anxiety, depression), and a variety of physiologic effects (such as decreased appetite, headaches), and more.
The two most commonly used scales to assess withdrawal symptoms include the 16‐item marijuana withdrawal checklist (MWC) and the 19‐item cannabis withdrawal scale (CWS).
Cannabis Withdrawal Treatment
Currently, treatment of cannabis withdrawal generally involves supportive counseling and psychoeducation regarding the typical symptoms, such as symptom time course, craving management, and supportive lifestyle interventions such as sleep hygiene and exercise (Connor et al., 2022).
Pharmacotherapy for Cannabis Withdrawal (Connor et al., 2022)
Currently, the data for pharmacological management of withdrawal symptoms are weak, and there are no FDA-approved medications at this time.
There have been at least 19 placebo-controlled studies, with some efficacy demonstrated for various pharmacological agents to target symptom relief, such as gabapentin, Ambien, and quetiapine for sleep disturbance, synthetic CB1 agonists nabiximol, as well as oral THC.
There are also ongoing studies of FAAH inhibitors which function to elevate endogenous anandamide levels for withdrawal symptoms and to reduce use (D'Souza et al., 2019), and some data show promise for high dose CBD (400-800mg) in mildly reducing overall cannabis use (Freeman et al., 2021).
There is also low-quality evidence of using the antioxidant supplement N-Acetylcysteine (NAC), which costs less than $1/day, for cannabis cravings (Sharma et al., 2022). Doses typically start at 600 mg once or twice daily then increase to 1200 mg twice daily as tolerated, with side effects mainly being GI related (nausea, diarrhea, abdominal pain), which can be mitigated if taken with food or split into three doses throughout the day.
Again, the current data is relatively weak and there are currently no FDA-approved pharmacotherapies for cannabis use disorder or withdrawal.
Psychotherapy Approaches to Cannabis Use Disorder
Forming a Therapeutic Alliance
A therapeutic alliance is central to addressing ambivalence around cannabis use. Rather than confronting or pressuring patients, we can help patients clarify what they want for themselves and explore how current behaviors may support or undermine these goals. This collaborative process increases openness, expressivity, and insight. For example, a patient may share a desire to save money for an apartment while spending $20 a day on cannabis and noticing diminished work motivation. By reflecting this discrepancy empathetically, we can highlight the tension between values and actions in a way that invites the patient to consider change.
Attachment to Cannabis
It is also important to recognize that cannabis is not simply a substance for many patients—it often carries emotional, relational, and ritualistic meaning. This attachment can be difficult to let go, and doing so may bring anxiety and a grieving process. Patients may need to mourn aspects of their relationship with cannabis while developing new, healthier sources of comfort, meaning, and connection. We can help by naming this process, by validating the emotions associated with this loss, and by discussing new coping strategies and rituals to replace cannabis.
Harm Reduction Approach
Using harm reduction approaches can diminish the negative impacts of cannabis use. Canadian guidelines offer advice on lower-risk cannabis use and addiction medicine providers emphasize practical steps:
Delaying use until adulthood
Keeping cannabis for the weekend
In terms of safety: edibles (in measured doses) > vaping flower > smoking flower > vaping concentrates
Count your hits (don’t smoke the whole joint)
Know the % THC, stick to low potency strains that include CBD if possible
Trusted sources, such as dispensaries
Organic, if possible (pesticide regulations vary from state to state)
Physical Risks Of Cannabis
When a client or a patient walks into your office and asks about cannabis and whether they should start it and/or continue its usage, of course all of its effects on the psyche and mind are of great importance, but it is additionally beneficial for us a clinicians to be familiar with the effects of cannabis on the physical body. Thus, here are some big takeaways that are important for clinicians and patients alike to be familiar with when considering cannabis usage:
Cannabis Hyperemesis Syndrome
Cannabis hyperemesis syndrome is an underrecognized syndrome of intractable cyclic vomiting seen in some long-term users of cannabis. Symptoms are frequently refractory to standard antiemetic therapies and accompanied by abdominal pain (Pergolizzi et al., 2018). The Rome IV criteria of CHS requires cannabinoid use and persistence of N/V symptoms for at least the past 6 months.
Mechanism: Prolonged high doses of Δ9-tetrahydrocannabinol (THC) act on the cannabinoid-1 receptor, resulting in changes to the endocannabinoid system primarily through CB1 receptor desensitization and internalization in the brainstem and enteric nervous system. This impairs normal anti-emetic signaling and paradoxically enhances emetogenic pathways, including increased activity of dopamine, serotonin, and substance P. These changes dysregulate stress and anxiety responses, thermoregulation, the transient receptor potential vanilloid system, and several neurotransmitter systems, and thus may mediate the pathophysiology of CHS (DeVuono & Parker, 2020).
Treatments: Per a 2022 meta review, the cessation of marijuana is the only definitive treatment of cannabis hyperemesis syndrome. Other treatments which have been found to provide some relief include hot water hydrotherapy, topical capsaicin cream, haloperidol, droperidol, benzodiazepines, propranolol, and aprepitant administration, with topical capsaicin cream and haloperidol being the only two medications whose efficacy was supported by RCTs (Senderovich et al., 2022).
The co-localized TRPV1 receptor system—implicated in thermoregulation and emesis modulation—may provide a compensatory pathway for the dysregulated endocannabinoid system, explaining the transient symptom relief observed with hot showers and topical capsaicin. THC’s lipophilicity and accumulation in adipose tissue further contribute to symptom relapses through stress- or fasting-induced lipolysis, re-releasing THC and reactivating emetic circuits. Genetic variants in TRPV1, CYP2C9, and DRD2 may increase susceptibility by altering THC metabolism, receptor sensitivity, or neurochemical balance (Loganatha et al., 2024).
With the legalization of marijuana, cannabis hyperemesis syndrome is bound to become more common and clinicians would be wise to have this diagnosis on their radar in marijuana users, avoiding unnecessary diagnostic tests when appropriate.
Cannabis and Cardiac Risks
Another area of question for many is the effects that cannabis may have on the cardiovascular system. One population-based, cross-sectional study of 2016 to 2020 data of 434,104 participants in the United States assessing the association of cannabis use with self-reported cardiovascular outcomes in multivariable regression models, adjusting for tobacco use and other characteristics in adults 18 to 74 years old.
Daily cannabis use was associated with myocardial infarction (aOR 1.25, CI 1.7-1.46) and stroke (aOR 1.42, CI 1.20-1.68). Among people who had never smoked tobacco, daily cannabis use was also associated with myocardial infarction (aOR 1.49, CI 1.03-2.15), stroke (aOR 2.16, CI 1.43-3.25). However, this study was limited by measuring its variables by self-report, as the outcomes could be subject to recall bias, and the self-reported cannabis use was in poor agreement with biochemical validation (Jeffers et al., 2024).
One prospective study of 22 young, healthy males prospectively examining the effects of THC-predominant cannabis and CBD-predominant cannabis found an increase in heart rate with smoking THC-predominant cannabis (Δ17±15 bpm, P<0.0001) and vaporizing THC‐predominant cannabis (V‐THC: Δ16±16 bpm; P<0.0001). An increase in mean arterial pressure was also observed (S‐THC: Δ7±6 mm Hg, V‐THC: Δ5±5 mm Hg; P<0.0001). These results were not seGenetic variants in TRPV1, CYP2C9, and DRD2 may increase susceptibility by altering THC metabolism, receptor sensitivity, or neurochemical balance with cannabidiol-predominant cannabis (Cheung et al., 2024).
When we evaluate clinically-relevant outcomes, however, the largest prospective study to date - the CARDIA study - failed to show an association between lifetime or recent cannabis use and cardiovascular events. There is suggestion on a molecular level and based on retrospective analyses that cannabis may have a negative impact on the cardiovascular system, but prospective clinical data has not confirmed these suggested findings. Further research is needed to better elucidate the association, if any, between cannabis and cardiovascular disease (Ghosh & Naderi, 2019).
Cannabis and Lung Disease
There has been extensive research on the effects of cannabis on the pulmonary system both in the acute and long-term settings.
To highlight a few of the biggest takeaways:
Unlike smoking tobacco, which can cause reflexive bronchoconstriction acutely, smoking cannabis results in temporary bronchodilation. Studies have shown that smoking marijuana with 2% THC by weight has been found to produce acute bronchodilation (Tashkin et al., 1973). In patients with rapid onset mild asthma, methacholine-induced bronchospasm, and exercise-induced bronchospasm as well, smoking a 2% THC marijuana cigarette reversed the bronchoconstriction (Tashkin et al., 1974; Tashkin et al., 1975). In fact, marijuana was used as a treatment for asthma in the 19th century (Grinspoon, 1969).
However, over the long term:
Young otherwise healthy heavy smokers of marijuana have increased prevalence of wheezing (25-37%), chronic cough (18 to 24%), sputum production (20 to 26%), and increased prevalence of at least one prolonged acute bronchitic episode over a retrospective 3-year period (10 to 14%) compared to nonsmokers (p < 0.05, chi square) (Tashkin et al., 1987).
An age-matched retrospective case-control chi-squared analysis evaluating chest CT examination results demonstrated higher rates of emphysema among marijuana smokers than nonsmokers and tobacco-only smokers (P < .001, P=0.006), and higher rates of bronchial thickening, bronchiectasis, and mucoid impaction compared with tobacco-only smokers (P = 0.009). This result was also observed when comparing marijuana smokers to non-smokers prior to age-matching (P < 0.001) (Murtha et al., 2022).
In a retrospective cohort study of Kaiser Permanente, the medical charts of patients in Oakland and San Francisco, CA, daily marijuana smokers have an increased risk for requiring outpatient visits for respiratory illnesses compared to non-smokers (RR = 1.19, CI = 1.01, 1.41) (Polen et al., 1993).
Cannabis and Lung Cancer
Many studies have explored the relationship between cannabis use and lung cancer, but results have been inconclusive. A 2013 prospective (n = 44,284) found that “heavy” cannabis smoking (defined as > 50 lifetime uses of marijuana) was significantly associated with an increased risk for lung cancer (hazard ratio 2.12, 95 % CI 1.08–4.14) developed over a 40-year period, even after adjusting for baseline tobacco use, alcohol use, respiratory conditions, and socioeconomic status (Callaghan et al., 2013). However, several other large studies have concluded that there is no relationship between cannabis use and lung cancer (Chatkin et al., 2017). Interestingly, one study used Mendelian randomization to look at 7 single nucleotide polymorphisms for > 180,000 individuals and use this to assess the effect of the genetic liability to lifetime cannabis use/CUD on pulmonary function and lung cancer (independent from tobacco smoking). The genetic liability was found to be associated with increased risk of squamous cell carcinoma (OR = 1.22, 95%, confidence interval = 1.07–1.39, p value = 0.003, q value = 0.025) (Baumeister et al., 2021).
Cannabis And Sex Hormones and Fertility
Sex Hormones
A retrospective cross-sectional study was performed using medical records data of 153 infertile men aged 18–59 years from 2009 to 2017 and used ANOVA to determine which habit or lifestyle factors (tobacco use, daily alcohol consumption, marijuana use, and sedentary lifestyle) could influence specific sperm parameters or hormone levels the most. Age (> or equal to 45 years) and clinical varicocele were accounted for as predictors. ANOVA for association b/w semen/hormonal parameters and epidemiological/clinical characteristics. Marijuana use was inversely related to serum estradiol and demonstrated the most substantial effect out of the lifestyle factors measured (Table 4: ß = −0.35, p < 0.01). Marijuana use was positively associated with prolactin levels (Table 5: ß = 0.30; p < 0.01) and sex hormone binding globulin (Table 7: ß = 0.19; p = 0.02). The study was limited by lack of a matched control group of fertile men (Teixeira et al., 2022).
Female Fertility
The enzymes involved in the endocannabinoids system are important for successful implantation of the embryo and human fertility. Estrogen and progesterone are also involved in maintaining endocannabinoid levels. Defects in this system can result in fetal loss. THC binds to the same receptors as endogenous cannabinoids (Battista et al., 2007; Battista et al., 2008; Taylor et al., 2007).
CB1 is important for oviduct transport and embryo development (Paria et al., 2001; Wang et al., 2004). Knocking out CB1 in mice showed pregnancy loss. CB1 deficiency resulted in retention of the embryo and ectopic pregnancy, demonstrating the importance of correct expression of CB1 for normal pregnancy. The endogenous levels of anandamide (AEA), which binds to CB1, are tightly regulated from the very beginning of pregnancy and are important for the timing of embryo implantation; any deviation from normal regulation severely compromises the pregnancy outcome.
THC, unlike endogenous ligands, is slowly metabolized and accumulates in fat deposits within the body and may mimic situations where an excess of endocannabinoids is produced or when re-uptake or removal of endogenous ligands is impaired (Schuel & Burkman, 2006).
Male Fertility
AEA has also been shown to depress motility and capacitation of human spermatozoa (Rossato et al., 2005).
A 2021 literature review found multiple studies both in animal studies and studies in infertile males which demonstrated that THC exposure leads to arrest in the production of sperm and spermatogenesis (Ryan et al., 2021). These results were corroborated cohort study of 18-28 year old Danish men evaluating the effects of marijuana usage on sperm concentration and total sperm count, as smoking more than once per week was associated with a 28% reduction in sperm concentration (95% confidence interval (CI): -48, -1), and a 29% lower total sperm count (95% CI: -46, -1) after adjustment for confounders (Gundersen et al., 2021).
Summary
Cannabis use is increasing at unprecedented rates, especially among those with depression and anxiety.
Depression: Cannabis users have higher odds of depression, particularly with heavy or adolescent-onset use; withdrawal-related dysphoria may perpetuate depressive cycles.
Suicidality: Adolescent and heavy cannabis use are linked to increased suicidal ideation and attempts, though confounding factors weaken causal claims.
Anxiety: Evidence for cannabis causing anxiety disorders is weak; short-term symptom relief is common, but there is no lasting benefit.
PTSD: Despite patient reports of benefit, RCTs and systematic reviews show no long-term improvement; CUD predicts poorer PTSD treatment outcomes.
Sleep: Cannabis may reduce sleep latency and nightmares acutely, but tolerance and withdrawal disrupt sleep; overall evidence is inconsistent.
CBD/Terpenes: Limited and mixed evidence that CBD or terpenes relieve anxiety or mitigate THC’s anxiogenic effects.
Cognition/IQ: Frequent use causes small, reversible deficits, but persistent adolescent-onset use is linked to lasting IQ decline (~6–8 points) and poorer life outcomes. Whether this is a direct effect of cannabis is unclear.
Cannabis Use Disorder: 8–19% of users meet criteria; withdrawal is common but usually mild; no FDA-approved treatments—psychotherapy and harm reduction are mainstays. If a patient is using cannabis, discuss their use nonjudgmentally to understand together what their goals are and see how cannabis might be an obstacle to achieving them. If they’re using relatively frequently, try cutting down and see what happens with their mood, energy levels, sleep, etc.
Physical risks: Includes cannabis hyperemesis syndrome, possible cardiovascular harms, pulmonary disease (chronic bronchitis, emphysema), and negative impacts on fertility/sex hormones.
References
Agrawal, A., Nelson, E. C., Bucholz, K. K., Tillman, R., Grucza, R. A., Statham, D. J., Madden, P. A., Martin, N. G., Heath, A. C., & Lynskey, M. T. (2017). Major depressive disorder, suicidal thoughts and behaviours, and cannabis involvement in discordant twins: a retrospective cohort study. The lancet. Psychiatry, 4(9), 706–714. https://doi.org/10.1016/S2215-0366(17)30280-8
Ahmed, S., Roth, R. M., Stanciu, C. N., & Brunette, M. F. (2021). The impact of THC and CBD in schizophrenia: A systematic review. Frontiers in Psychiatry, 12, 694394. https://doi.org/10.3389/fpsyt.2021.694394
Albaugh, M. D., Ottino-Gonzalez, J., Sidwell, A., Lepage, C., Juliano, A., Owens, M. M., Chaarani, B., Spechler, P., Fontaine, N., Rioux, P., Lewis, L., Jeon, S., Evans, A., D’Souza, D., Radhakrishnan, R., Banaschewski, T., Bokde, A. L. W., Quinlan, E. B., Conrod, P., ... Garavan, H. (2021). Association of cannabis use during adolescence with neurodevelopment. JAMA Psychiatry. Advance online publication. https://doi.org/10.1001/jamapsychiatry.2021.1258
Albaugh, M. D., Owens, M. M., Juliano, A., Ottino-Gonzalez, J., Cupertino, R., Cao, Z., Mackey, S., Lepage, C., Rioux, P., Evans, A., Banaschewski, T., Bokde, A. L. W., Conrod, P., Desrivières, S., Flor, H., Grigis, A., Gowland, P., Heinz, A., Ittermann, B., … IMAGEN Consortium. (2023). Differential associations of adolescent versus young adult cannabis initiation with longitudinal brain change and behavior. Molecular Psychiatry, 28(11), 5173–5182. https://doi.org/10.1038/s41380-023-02148-2
Alcover, K. C., & Thompson, C. L. (2020). Patterns of Mean Age at Drug Use Initiation Among Adolescents and Emerging Adults, 2004-2017. JAMA pediatrics, 174(7), 725–727. https://doi.org/10.1001/jamapediatrics.2019.6235
Amaral, C., Carvalho, C., Scaranelo, A., Chapman, K., Chatkin, J., & Ferreira, I. (2023). Cannabis and sleep disorders: not ready for prime time? A qualitative scoping review. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine, 19(5), 975–990. https://doi.org/10.5664/jcsm.10428
Anderson, L. L., Doohan, P. T., Oldfield, L., Kevin, R. C., Arnold, J. C., Berger, M., Amminger, G. P., & McGregor, I. S. (2021). Citalopram and cannabidiol: In vitro and in vivo evidence of pharmacokinetic interactions relevant to the treatment of anxiety disorders in young people. Journal of Clinical Psychopharmacology, 41(5), 525-533. https://doi.org/10.1097/JCP.0000000000001427
Arkell, T. R., Kevin, R. C., Vinckenbosch, F., Lintzeris, N., Theunissen, E., Ramaekers, J. G., & McGregor, I. S. (2022). Sex differences in acute cannabis effects revisited: Results from two randomized, controlled trials. Addiction biology, 27(2), e13125. https://doi.org/10.1111/adb.13125
Bahji, A., Stephenson, C., Tyo, R., Hawken, E. R., & Seitz, D. P. (2020). Prevalence of Cannabis Withdrawal Symptoms Among People With Regular or Dependent Use of Cannabinoids: A Systematic Review and Meta-analysis. JAMA network open, 3(4), e202370. https://doi.org/10.1001/jamanetworkopen.2020.2370
Baldaçara, L., Ramos, A., & Castaldelli-Maia, J. M. (2023). Managing drug-induced psychosis. International Review of Psychiatry, 35(5–6), 496–502. https://doi.org/10.1080/09540261.2023.2261544
Battistella, G., Fornari, E., Annoni, J. M., Chtioui, H., Dao, K., Fabritius, M., Favrat, B., Mall, J. F., Maeder, P., & Giroud, C. (2014). Long-term effects of cannabis on brain structure. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology, 39(9), 2041–2048. https://doi.org/10.1038/npp.2014.67
Bedard-Gilligan, M., Garcia, N., Zoellner, L. A., & Feeny, N. C. (2018). Alcohol, cannabis, and other drug use: Engagement and outcome in PTSD treatment. Psychology of Addictive Behaviors, 32(3), 277–288. https://doi.org/10.1037/adb0000355
Benyamina, A., Kebir, O., Blecha, L., Reynaud, M., & Krebs, M.-O. (2011). CNR1 gene polymorphisms in addictive disorders: A systematic review and a meta-analysis. Addiction Biology, 16(1), 1–6. https://doi.org/10.1111/j.1369-1600.2009.00198.x
Beyer, E., Poudel, G., Antonopoulos, S., Thomson, H., & Lorenzetti, V. (2024). Brain reward function in people who use cannabis: A systematic review. Frontiers in Behavioral Neuroscience, 17, 1323609. https://doi.org/10.3389/fnbeh.2023.1323609
Bhat, T. A., Kalathil, S. G., Goniewicz, M. L., Hutson, A., & Thanavala, Y. (2023). Not all vaping is the same: differential pulmonary effects of vaping cannabidiol versus nicotine. Thorax, 78(9), 922–932. https://doi.org/10.1136/thorax-2022-218743
Blanco, C., Hasin, D. S., Wall, M. M., Flórez-Salamanca, L., Hoertel, N., Wang, S., Kerridge, B. T., & Olfson, M. (2016). Cannabis use and risk of psychiatric disorders: Prospective evidence from a US national longitudinal study. JAMA Psychiatry, 73(4), 388–395. https://doi.org/10.1001/jamapsychiatry.2015.3229
Bolsoni, L. M., Crippa, J. A. S., Hallak, J. E. C., Guimarães, F. S., & Zuardi, A. W. (2022). The anxiolytic effect of cannabidiol depends on the nature of the trauma when patients with post-traumatic stress disorder recall their trigger event. Revista brasileira de psiquiatria (Sao Paulo, Brazil : 1999), 44(3), 298–307. https://doi.org/10.1590/1516-4446-2021-2317
Bonn-Miller, M. O., Boden, M. T., Vujanovic, A. A., & Drescher, K. D. (2013). Prospective investigation of the impact of cannabis use disorders on posttraumatic stress disorder symptoms among veterans in residential treatment. Psychological Trauma: Theory, Research, Practice, and Policy, 5(2), 193–200. https://doi.org/10.1037/a0026621
Bonn-Miller, M. O., Sisley, S., Riggs, P., Yazar-Klosinski, B., Wang, J. B., Loflin, M. J. E., Shechet, B., Hennigan, C., Matthews, R., & Emerson, A. (2021). The short-term impact of 3 smoked cannabis preparations versus placebo on PTSD symptoms: A randomized cross-over clinical trial. PLOS ONE, 16(3), e0246990. https://doi.org/10.1371/journal.pone.0246990
Bourque, J., & Potvin, S. (2021). Cannabis and cognitive functioning: From acute to residual effects, from randomized controlled trials to prospective designs. Frontiers in Psychiatry, 12, 596601. https://doi.org/10.3389/fpsyt.2021.596601
Calabrese, E. J., & Rubio-Casillas, A. (2018). Biphasic effects of THC in memory and cognition. European Journal of Clinical Investigation, 48, e12920. https://doi.org/10.1111/eci.12920
Caspi, A., Moffitt, T. E., Cannon, M., McClay, J., Murray, R., Harrington, H., Taylor, A., Arseneault, L., Williams, B., Braithwaite, A., Poulton, R., & Craig, I. W. (2005). Moderation of the effect of adolescent-onset cannabis use on adult psychosis by a functional polymorphism in the catechol-O-methyltransferase gene: Longitudinal evidence of a gene × environment interaction. Biological Psychiatry, 57(10), 1117–1127. https://doi.org/10.1016/j.biopsych.2005.01.026
Caulkins, J. P. (2024). Changes in self-reported cannabis use in the United States from 1979 to 2022. Addiction, 119(9), 1648–1652. https://doi.org/10.1111/add.16519
Centers for Disease Control and Prevention (CDC). (2024, February 22). Cannabis facts and stats. https://www.cdc.gov/cannabis/data-research/facts-stats/index.html
Cheetham, A., Allen, N. B., Whittle, S., Simmons, J. G., Yücel, M., & Lubman, D. I. (2012). Orbitofrontal volumes in early adolescence predict initiation of cannabis use: a 4-year longitudinal and prospective study. Biological psychiatry, 71(8), 684–692. https://doi.org/10.1016/j.biopsych.2011.10.029
Churchill, V., Chubb, C. S., Popova, L., Spears, C. A., & Pigott, T. (2025). The association between cannabis and depression: An updated systematic review and meta-analysis. Psychological Medicine, 55, e44. https://doi.org/10.1017/S0033291724003143
Churchwell, J. C., Lopez-Larson, M., & Yurgelun-Todd, D. A. (2010). Altered frontal cortical volume and decision making in adolescent cannabis users. Frontiers in psychology, 1, 225. https://doi.org/10.3389/fpsyg.2010.00225
Colizzi, M., Weltens, N., McGuire, P., Van Oudenhove, L., & Bhattacharyya, S. (2020). Delta-9-tetrahydrocannabinol increases striatal glutamate levels in healthy individuals: Implications for psychosis. Molecular Psychiatry, 25, 3231–3240. https://doi.org/10.1038/s41380-019-0374-8
Connor, J. P., Stjepanović, D., Budney, A. J., Le Foll, B., & Hall, W. D. (2022). Clinical management of cannabis withdrawal. Addiction (Abingdon, England), 117(7), 2075–2095. https://doi.org/10.1111/add.15743
Coughlin, L. N., Bonar, E. E., Bohnert, K. M., Jannausch, M., Walton, M. A., Blow, F. C., & Ilgen, M. A. (2019). Changes in urban and rural cigarette smoking and cannabis use from 2007 to 2017 in adults in the United States. Drug and Alcohol Dependence, 205, 107699. https://doi.org/10.1016/j.drugalcdep.2019.107699
Cougle, J. R., Hakes, J. K., Macatee, R. J., Chavarria, J., & Zvolensky, M. J. (2015). Quality of life and risk of psychiatric disorders among regular users of alcohol, nicotine, and cannabis: An analysis of the National Epidemiological Survey on Alcohol and Related Conditions (NESARC). Journal of Psychiatric Research, 66–67, 135-141. https://doi.org/10.1016/j.jpsychires.2015.05.004
Cuttler, C., Spradlin, A., & McLaughlin, R. J. (2018). A naturalistic examination of the perceived effects of cannabis on negative affect. Journal of Affective Disorders, 235, 198–205. https://doi.org/10.1016/j.jad.2018.04.054
Davis, J. P., Pedersen, E. R., Tucker, J. S., Prindle, J., Dunbar, M. S., Rodriguez, A., Seelam, R., & D’Amico, E. J. (2022). Directional associations between cannabis use and anxiety symptoms from late adolescence through young adulthood. Drug and Alcohol Dependence, 241, 109704. https://doi.org/10.1016/j.drugalcdep.2022.109704
Di Forti, M., Iyegbe, C., Sallis, H., Kolliakou, A., Falcone, M. A., Paparelli, A., Sirianni, M., La Cascia, C., Stilo, S. A., Marques, T. R., Handley, R., Mondelli, V., Dazzan, P., Pariante, C., David, A. S., Morgan, C., Powell, J., & Murray, R. M. (2012). Confirmation that the AKT1 (rs2494732) genotype influences the risk of psychosis in cannabis users. Biological Psychiatry, 72(10), 811–816. https://doi.org/10.1016/j.biopsych.2012.06.020
Di Forti, M., Quattrone, D., Freeman, T. P., Tripoli, G., Gayer-Anderson, C., Quigley, H., Rodriguez, V., Jongsma, H. E., Ferraro, L., La Cascia, C., La Barbera, D., Tarricone, I., Berardi, D., Szöke, A., Arango, C., Tortelli, A., Velthorst, E., Bernardo, M., Marta Del-Ben, C., ... Murray, R. M. (2019). The contribution of cannabis use to variation in the incidence of psychotic disorder across Europe (EU-GEI): A multicentre case-control study. The Lancet Psychiatry, 6(5), 427–436. https://doi.org/10.1016/S2215-0366(19)30048-3
D'Souza, D. C., Perry, E., MacDougall, L., Ammerman, Y., Cooper, T., Wu, Y. T., Braley, G., Gueorguieva, R., & Krystal, J. H. (2004). The psychotomimetic effects of intravenous delta-9-tetrahydrocannabinol in healthy individuals: Implications for psychosis. Neuropsychopharmacology, 29(8), 1558–1572. https://doi.org/10.1038/sj.npp.1300496
D’Souza, D. C., Abi-Saab, W. M., Madonick, S., Forselius-Bielen, K., Doersch, A., Braley, G., Gueorguieva, R., Cooper, T. B., & Krystal, J. H. (2005). Delta-9-tetrahydrocannabinol effects in schizophrenia: Implications for cognition, psychosis, and addiction. Biological Psychiatry, 57(6), 594–608. https://doi.org/10.1016/j.biopsych.2004.12.006
D’Souza, D. C., Cortes-Briones, J. A., Ranganathan, M., Thurnauer, H., Creatura, G., Surti, T., Planeta, B., Neumeister, A., Pittman, B., Normandin, M. D., Kapinos, M., Ropchan, J., Huang, Y., Carson, R. E., & Skosnik, P. D. (2019). Efficacy and safety of a fatty acid amide hydrolase inhibitor (PF-04457845) in the treatment of cannabis withdrawal and dependence in men: A double-blind, placebo-controlled, parallel group, phase 2a single-site randomised controlled trial. The Lancet Psychiatry, 6(1), 35–45. https://doi.org/10.1016/S2215-0366(18)30427-9
Englund, A., Morrison, P. D., Nottage, J., Hague, D., Kane, F., Bonaccorso, S., Stone, J. M., Reichenberg, A., Brenneisen, R., Holt, D., Feilding, A., Walker, L., Murray, R. M., & Kapur, S. (2013). Cannabidiol inhibits THC-elicited paranoid symptoms and hippocampal-dependent memory impairment. Journal of psychopharmacology (Oxford, England), 27(1), 19–27. https://doi.org/10.1177/0269881112460109
Feingold, D., Weiser, M., Rehm, J., & Lev-Ran, S. (2016). The association between cannabis use and anxiety disorders: Results from a population-based representative sample. European Neuropsychopharmacology, 26(3), 493-505. https://doi.org/10.1016/j.euroneuro.2015.12.037
Feingold, D., Rehm, J., & Lev-Ran, S. (2017). Cannabis use and the course and outcome of major depressive disorder: A population-based longitudinal study. Psychiatry Research, 251, 225–234. https://doi.org/10.1016/j.psychres.2017.02.027
Filbey, F. M., Aslan, S., Calhoun, V. D., Spence, J. S., Damaraju, E., Caprihan, A., & Segall, J. (2014). Long-term effects of marijuana use on the brain. Proceedings of the National Academy of Sciences of the United States of America, 111(47), 16913–16918. https://doi.org/10.1073/pnas.1415297111
Fiorentini, A., Cantù, F., Crisanti, C., Cereda, G., Oldani, L., & Brambilla, P. (2021). Substance-induced psychoses: An updated literature review. Frontiers in Psychiatry, 12, 694863. https://doi.org/10.3389/fpsyt.2021.694863
Freeman, D., Dunn, G., Murray, R. M., Evans, N., Lister, R., Antley, A., Slater, M., Godlewska, B., Cornish, R., Williams, J., Di Simplicio, M., Igoumenou, A., Brenneisen, R., Tunbridge, E. M., Harrison, P. J., Harmer, C. J., Cowen, P., & Morrison, P. D. (2015). How cannabis causes paranoia: Using the intravenous administration of Δ9-tetrahydrocannabinol (THC) to identify key cognitive mechanisms leading to paranoia. Schizophrenia Bulletin, 41(2), 391–399. https://doi.org/10.1093/schbul/sbu098
Gage, S. H., Jones, H. J., Burgess, S., Bowden, J., Davey Smith, G., Zammit, S., & Munafò, M. R. (2017). Assessing causality in associations between cannabis use and schizophrenia risk: a two-sample Mendelian randomization study. Psychological Medicine, 47(5), 971–980. doi:10.1017/S0033291716003172
Gali, K., Winter, S. J., Ahuja, N. J., Frank, E., & Prochaska, J. J. (2021). Changes in cannabis use, exposure, and health perceptions following legalization of adult recreational cannabis use in California: a prospective observational study. Substance abuse treatment, prevention, and policy, 16(1), 16. https://doi.org/10.1186/s13011-021-00352-3
Garey, L., Olofsson, H., Garza, T., Rogers, A. H., Kauffman, B. Y., & Zvolensky, M. J. (2020). Directional effects of anxiety and depressive disorders with substance use: A review of recent prospective research. Current Addiction Reports, 7(4), 344–355. https://doi.org/10.1007/s40429-020-00321-z
Gillespie, N. A., Pasman, J. A., Treur, J. L., Derks, E. M., Verweij, K. J. H., & Vink, J. M. (2019). High-potency cannabis and incident psychosis: Correcting the causal assumption. The Lancet Psychiatry, 6(6), 464. https://doi.org/10.1016/S2215-0366(19)30174-9
Gilman, J. M., Schuster, R. M., Potter, K. W., Schmitt, W., Wheeler, G., Pachas, G. N., Hickey, S., Cooke, M. E., Dechert, A., Plummer, R., Tervo-Clemmens, B., Schoenfeld, D. A., & Evins, A. E. (2022). Effect of medical marijuana card ownership on pain, insomnia, and affective disorder symptoms in adults: A randomized clinical trial. JAMA Network Open, 5(3), e222106. https://doi.org/10.1001/jamanetworkopen.2022.2106
Glodosky, N. C., Cuttler, C., & McLaughlin, R. J. (2021). A review of the effects of acute and chronic cannabinoid exposure on the stress response. Frontiers in neuroendocrinology, 63, 100945. https://doi.org/10.1016/j.yfrne.2021.100945
Gobbi, G., Atkin, T., Zytynski, T., Wang, S., Askari, S., Boruff, J., Ware, M., Marmorstein, N., Cipriani, A., Dendukuri, N., & Mayo, N. (2019). Association of Cannabis Use in Adolescence and Risk of Depression, Anxiety, and Suicidality in Young Adulthood: A Systematic Review and Meta-analysis. JAMA psychiatry, 76(4), 426–434. https://doi.org/10.1001/jamapsychiatry.2018.4500
Gorfinkel, L. R., Stohl, M., & Hasin, D. (2020). Association of Depression With Past-Month Cannabis Use Among US Adults Aged 20 to 59 Years, 2005 to 2016. JAMA network open, 3(8), e2013802. https://doi.org/10.1001/jamanetworkopen.2020.13802
Grant, B. F., Goldstein, R. B., Saha, T. D., Chou, S. P., Jung, J., Zhang, H., Pickering, R. P., Ruan, W. J., Smith, S. M., Huang, B., & Hasin, D. S. (2015). Epidemiology of DSM-5 Alcohol Use Disorder: Results From the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA psychiatry, 72(8), 757–766. https://doi.org/10.1001/jamapsychiatry.2015.0584
Gunasekaran, N., Long, L. E., Dawson, B. L., Hansen, G. H., Richardson, D. P., Li, K. M., Arnold, J. C., & McGregor, I. S. (2009). Reintoxication: the release of fat-stored delta(9)-tetrahydrocannabinol (THC) into blood is enhanced by food deprivation or ACTH exposure. British journal of pharmacology, 158(5), 1330–1337. https://doi.org/10.1111/j.1476-5381.2009.00399.x
Hamilton, I., & Monaghan, M. (2019). Cannabis and psychosis: Are we any closer to understanding the relationship? Current Psychiatry Reports, 21(48). https://doi.org/10.1007/s11920-019-1044-x
Hammond, C. J., Allick, A., Park, G., Rizwan, B., Kim, K., Lebo, R., Nanavati, J., Parvaz, M. A., & Ivanov, I. (2022). A Meta-Analysis of fMRI Studies of Youth Cannabis Use: Alterations in Executive Control, Social Cognition/Emotion Processing, and Reward Processing in Cannabis Using Youth. Brain Sciences, 12(10), 1281. https://doi.org/10.3390/brainsci12101281
Harding, I. H., Solowij, N., Harrison, B. J., Takagi, M., Lorenzetti, V., Lubman, D. I., Seal, M. L., Pantelis, C., & Yücel, M. (2012). Functional connectivity in brain networks underlying cognitive control in chronic cannabis users. Neuropsychopharmacology, 37(7), 1923–1933. https://doi.org/10.1038/npp.2012.39
Hasin D. S. (2018). US Epidemiology of Cannabis Use and Associated Problems. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology, 43(1), 195–212. https://doi.org/10.1038/npp.2017.198
Hill, M. N., Haney, M., Hillard, C. J., Karhson, D. S., & Vecchiarelli, H. A. (2023). The endocannabinoid system as a putative target for the development of novel drugs for the treatment of psychiatric illnesses. Psychological medicine, 53(15), 7006–7024. https://doi.org/10.1017/S0033291723002465
Hill, M. L., Kline, A. C., Saraiya, T. C., Gette, J., Ruglass, L. M., Norman, S. B., Back, S. E., Saavedra, L. M., Hien, D. A., & Morgan-López, A. A. (2024b). Cannabis use and trauma-focused treatment for co-occurring posttraumatic stress disorder and substance use disorders: A meta-analysis of individual patient data. Journal of Anxiety Disorders, 102, 102827. https://doi.org/10.1016/j.janxdis.2024.102827
Hill, M., Loflin, M., Hicks, T., Browne, K., & Norman, S. D. (2024a). Marijuana use and posttraumatic stress disorder (PTSD) in veterans. U.S. Department of Veterans Affairs. https://www.ptsd.va.gov/professional/treat/cooccurring/marijuana_ptsd_vets.asp
Hillard C. J. (2015). The Endocannabinoid Signaling System in the CNS: A Primer. International review of neurobiology, 125, 1–47. https://doi.org/10.1016/bs.irn.2015.10.001
Hindocha, C., Freeman, T. P., Schafer, G., Gardener, C., Das, R. K., Morgan, C. J., & Curran, H. V. (2015). Acute effects of delta-9-tetrahydrocannabinol, cannabidiol and their combination on facial emotion recognition: a randomised, double-blind, placebo-controlled study in cannabis users. European neuropsychopharmacology : the journal of the European College of Neuropsychopharmacology, 25(3), 325–334. https://doi.org/10.1016/j.euroneuro.2014.11.014
Hughes, J. R., Fingar, J. R., Budney, A. J., Naud, S., Helzer, J. E., & Callas, P. W. (2014). Marijuana use and intoxication among daily users: an intensive longitudinal study. Addictive behaviors, 39(10), 1464–1470. https://doi.org/10.1016/j.addbeh.2014.05.024
Hutchison, K. E., Bidwell, L. C., Ellingson, J. M., & Bryan, A. D. (2019). Cannabis and Health Research: Rapid Progress Requires Innovative Research Designs. Value in health : the journal of the International Society for Pharmacoeconomics and Outcomes Research, 22(11), 1289–1294. https://doi.org/10.1016/j.jval.2019.05.005
Jacobus, J., & Tapert, S. F. (2014). Effects of cannabis on the adolescent brain. Current pharmaceutical design, 20(13), 2186–2193. https://doi.org/10.2174/13816128113199990426
Jakowiecki, J., Abel, R., Orzeł, U., Pasznik, P., Preissner, R., & Filipek, S. (2021). Allosteric Modulation of the CB1 Cannabinoid Receptor by Cannabidiol—A Molecular Modeling Study of the N-Terminal Domain and the Allosteric-Orthosteric Coupling. Molecules, 26(9), 2456. https://doi.org/10.3390/molecules26092456
Jang, S.-K., Saunders, G., Liu, M., Jiang, Y., Liu, D. J., & Vrieze, S. (2022). Genetic correlation, pleiotropy, and causal associations between substance use and psychiatric disorder. Psychological Medicine, 52(5), 968–978. https://doi.org/10.1017/S003329172000272X
Jefsen, O. H., Erlangsen, A., Nordentoft, M., & Hjorthøj, C. (2023). Cannabis Use Disorder and Subsequent Risk of Psychotic and Nonpsychotic Unipolar Depression and Bipolar Disorder. JAMA psychiatry, 80(8), 803–810. https://doi.org/10.1001/jamapsychiatry.2023.1256
Johnson E. C., Hatoum A. S., Deak J. D., Polimanti R., Murray R. M., Edenberg H. J., Gelernter J., Di Forti M., and Agrawal A. (2021) The relationship between cannabis and schizophrenia: a genetically informed perspective. Addiction, 116: 3227–3234. https://doi.org/10.1111/add.15534
Karniol, I. G., Shirakawa, I., Kasinski, N., Pfeferman, A., & Carlini, E. A. (1974). Cannabidiol interferes with the effects of delta 9 - tetrahydrocannabinol in man. European journal of pharmacology, 28(1), 172–177. https://doi.org/10.1016/0014-2999(74)90129-0
Karschner, E. L., Darwin, W. D., McMahon, R. P., Liu, F., Wright, S., Goodwin, R. S., & Huestis, M. A. (2011). Subjective and physiological effects after controlled Sativex and oral THC administration. Clinical pharmacology and therapeutics, 89(3), 400–407. https://doi.org/10.1038/clpt.2010.318
Kearn, C. S., Greenberg, M. J., DiCamelli, R., Kurzawa, K., & Hillard, C. J. (1999). Relationships between ligand affinities for the cerebellar cannabinoid receptor CB1 and the induction of GDP/GTP exchange. Journal of neurochemistry, 72(6), 2379–2387. https://doi.org/10.1046/j.1471-4159.1999.0722379.x
Kraemer, M., Madea, B., & Hess, C. (2019). Detectability of various cannabinoids in plasma samples of cannabis users: Indicators of recent cannabis use? Drug Testing and Analysis, 11(10), 1498–1506. https://doi.org/10.1002/dta.2682
Kuepper, R., van Os, J., Lieb, R., Wittchen, H., Höfler, M., Henquet, C., & et al. (2011). Continued cannabis use and risk of incidence and persistence of psychotic symptoms: 10-year follow-up cohort study. BMJ, 342, d738. https://doi.org/10.1136/bmj.d738
Kuepper, R., Ceccarini, J., Lataster, J., van Os, J., van Kroonenburgh, M., van Gerven, J. M. A., Marcelis, M., Van Laere, K., & Henquet, C. (2013). Delta-9-tetrahydrocannabinol-induced dopamine release as a function of psychosis risk: 18F-fallypride positron emission tomography study. PLOS ONE, 8(7), e70378. https://doi.org/10.1371/journal.pone.0070378
LaFrance, E. M., Glodosky, N. C., Bonn-Miller, M., & Cuttler, C. (2020). Short and Long-Term Effects of Cannabis on Symptoms of Post-Traumatic Stress Disorder. Journal of affective disorders, 274, 298–304. https://doi.org/10.1016/j.jad.2020.05.132
Langlois, C., Potvin, S., Khullar, A., & Tourjman, S. V. (2021). Down and High: Reflections Regarding Depression and Cannabis. Frontiers in psychiatry, 12, 625158. https://doi.org/10.3389/fpsyt.2021.625158
Lev-Ran, S., Roerecke, M., Le Foll, B., George, T. P., McKenzie, K., & Rehm, J. (2014). The association between cannabis use and depression: a systematic review and meta-analysis of longitudinal studies. Psychological medicine, 44(4), 797–810. https://doi.org/10.1017/S0033291713001438
Lichenstein S. D. (2022). THC, CBD, and Anxiety: A review of recent findings on the anxiolytic and anxiogenic effects of cannabis' primary cannabinoids. Current addiction reports, 9(4), 473–485. https://doi.org/10.1007/s40429-022-00450-7
Lopez-Larson, M. P., Bogorodzki, P., Rogowska, J., McGlade, E., King, J. B., Terry, J., & Yurgelun-Todd, D. (2011). Altered prefrontal and insular cortical thickness in adolescent marijuana users. Behavioural brain research, 220(1), 164–172. https://doi.org/10.1016/j.bbr.2011.02.001
Lorenzetti, V., Kowalczyk, M., Duehlmeyer, L., Greenwood, L. M., Chye, Y., Yücel, M., Whittle, S., & Roberts, C. A. (2023). Brain Anatomical Alterations in Young Cannabis Users: Is it All Hype? A Meta-Analysis of Structural Neuroimaging Studies. Cannabis and cannabinoid research, 8(1), 184–196. https://doi.org/10.1089/can.2021.0099
Love, C. A., Kim, H. H., Tallman, K. A., Clapp, P. W., Porter, N. A., & Jaspers, I. (2023). Vaping Induced Cannabidiol (CBD) Oxidation Product CBD Quinone Forms Protein Adducts with KEAP1 and Activates KEAP1-Nrf2 Genes. Chemical research in toxicology, 36(4), 565–569. https://doi.org/10.1021/acs.chemrestox.3c00038
Lucas, C. J., Galettis, P., & Schneider, J. (2018). The pharmacokinetics and the pharmacodynamics of cannabinoids. British Journal of Clinical Pharmacology, 84(11), 2477–2482. https://doi.org/10.1111/bcp.13710
Lucatch, A. M., Kloiber, S. M., Meyer, J. H., Rizvi, S. J., & George, T. P. (2020). Effects of extended cannabis abstinence in major depressive disorder. The Canadian Journal of Addiction, 11(3), 33-41. https://doi.org/10.1097/CXA.0000000000000090
Lutz B. (2020). Neurobiology of cannabinoid receptor signaling . Dialogues in clinical neuroscience, 22(3), 207–222. https://doi.org/10.31887/DCNS.2020.22.3/blutz
Masataka N. (2019). Anxiolytic Effects of Repeated Cannabidiol Treatment in Teenagers With Social Anxiety Disorders. Frontiers in psychology, 10, 2466. https://doi.org/10.3389/fpsyg.2019.02466
Matochik, J. A., Eldreth, D. A., Cadet, J.-L., & Bolla, K. I. (2005). Altered brain tissue composition in heavy marijuana users. Drug and Alcohol Dependence, 77(1), 23-30. https://doi.org/10.1016/j.drugalcdep.2004.06.011
Mayo Clinic Laboratories. (n.d.). Marijuana. Retrieved February 15, 2025, from https://news.mayocliniclabs.com/therapeutics/drug-class-testing/marijuana/
McCarthy, J. (2020, January 31). What percentage of americans smoke marijuana? Gallup.com. https://news.gallup.com/poll/284135/percentage-americans-smoke-marijuana.aspx
McGee, R., Williams, S., & Nada-Raja, S. (2005). Is cigarette smoking associated with suicidal ideation among young people? American Journal of Psychiatry, 162(3), 619–624. https://doi.org/10.1176/appi.ajp.162.3.619
Medina, K. L., McQueeny, T., Nagel, B. J., Hanson, K. L., Yang, T. T., & Tapert, S. F. (2009). Prefrontal cortex morphometry in abstinent adolescent marijuana users: subtle gender effects. Addiction biology, 14(4), 457–468. https://doi.org/10.1111/j.1369-1600.2009.00166.x
Meier, M. H., Caspi, A., Ambler, A., Harrington, H., Houts, R., Keefe, R. S., McDonald, K., Ward, A., Poulton, R., & Moffitt, T. E. (2012). Persistent cannabis users show neuropsychological decline from childhood to midlife. Proceedings of the National Academy of Sciences of the United States of America, 109(40), E2657–E2664. https://doi.org/10.1073/pnas.1206820109
Millar, S. A., Stone, N. L., Yates, A. S., & O'Sullivan, S. E. (2018). A Systematic Review on the Pharmacokinetics of Cannabidiol in Humans. Frontiers in pharmacology, 9, 1365. https://doi.org/10.3389/fphar.2018.01365
Miller, A. P., Baranger, D. A. A., Paul, S. E., Hatoum, A. S., Rogers, C., Bogdan, R., & Agrawal, A. (2023). Characteristics associated with cannabis use initiation by late childhood and early adolescence in the Adolescent Brain Cognitive Development (ABCD) Study. JAMA Pediatrics, 177(8), 861–863. https://doi.org/10.1001/jamapediatrics.2023.1801
Mørland, J., & Bramness, J. G. (2020). Δ9-tetrahydrocannabinol (THC) is present in the body between smoking sessions in occasional non-daily cannabis users. Forensic Science International, 309, 110188. https://doi.org/10.1016/j.forsciint.2020.110188
Myran, D. T., Harrison, L. D., Pugliese, M., Solmi, M., Anderson, K. K., Fiedorowicz, J. G., Perlman, C. M., Webber, C., Finkelstein, Y., & Tanuseputro, P. (2023). Transition to schizophrenia spectrum disorder following emergency department visits due to substance use with and without psychosis. JAMA Psychiatry, 80(11), 1169–1174. https://doi.org/10.1001/jamapsychiatry.2023.3582
Myran, D. T., Pugliese, M., Harrison, L. D., Solmi, M., Anderson, K. K., Fiedorowicz, J. G., Finkelstein, Y., Manuel, D., Taljaard, M., Webber, C., & Tanuseputro, P. (2025a). Changes in incident schizophrenia diagnoses associated with cannabis use disorder after cannabis legalization. JAMA Network Open, 8(2), e2457868. https://doi.org/10.1001/jamanetworkopen.2024.57868
Myran, D. T., Pugliese, M., McDonald, A. J., Xiao, J., Fischer, B., Finkelstein, Y., Tanuseputro, P., Firth, J., Pakpour, A., Hsu, C.-W., Chang, W.-C., & Solmi, M. (2025b). Cannabis use disorder emergency department visits and hospitalizations and 5-year mortality. JAMA Network Open, 8(2), e2457852. https://doi.org/10.1001/jamanetworkopen.2024.57852
Onaemo, V. N., Fawehinmi, T. O., & D'Arcy, C. (2021). Comorbid cannabis use disorder with major depression and generalized anxiety disorder: A systematic review with meta-analysis of nationally representative epidemiological surveys. Journal of Affective Disorders, 281, 467–475. https://doi.org/10.1016/j.jad.2020.12.043
Ousey, G. C., & Maume, M. O. (1997). The Grass is Always Greener: Explaining Rural and Urban Differences in Marijuana Use. Sociological Focus, 30(3), 295–305. https://doi.org/10.1080/00380237.1997.10571080
Pasman, J. A., Verweij, K. J. H., Gerring, Z., Stringer, S., Sanchez-Roige, S., Treur, J. L., Abdellaoui, A., Nivard, M. G., Baselmans, B. M. L., Ong, J.-S., Ip, H. F., van der Zee, M. D., Bartels, M., Day, F. R., Fontanillas, P., Elson, S. L., 23andMe Research Team, de Wit, H., Davis, L. K., MacKillop, J., ... Vink, J. M. (2018). GWAS of lifetime cannabis use reveals new risk loci, genetic overlap with psychiatric traits, and a causal effect of schizophrenia liability. Nature Neuroscience, 21(8), 1161–1170. https://doi.org/10.1038/s41593-018-0206-1
Patel, J., & Marwaha, R. (2024). Cannabis use disorder. In StatPearls. Treasure Island, FL: StatPearls Publishing. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK538131/
Patrick, M. E., Miech, R. A., Johnston, L. D., & O’Malley, P. M. (2024). Monitoring the Future Panel Study annual report: National data on substance use among adults ages 19 to 65, 1976–2023. Institute for Social Research, University of Michigan. https://monitoringthefuture.org/wp-content/uploads/2024/07/mtfpanel2024.pdf
Pauselli, L. (2018). Chapter 8 - Cannabis-induced psychotic disorders. In M. T. Compton & M. W. Manseau (Eds.), The complex connection between cannabis and schizophrenia (pp. 183–197). Academic Press. https://doi.org/10.1016/B978-0-12-804791-0.00008-2
Poyatos-Pedrosa, C., Bernabe-Valero, G., Pelacho-Ríos, L., & Iborra-Marmolejo, I. (2024). Cannabis and anhedonia: A systematic review. Psychiatry Research, 339, 116041. https://doi.org/10.1016/j.psychres.2024.116041
Puder, D. (Host). (2019, May 1). Marijuana and Mental Health (No. 44) [Audio podcast episode]. In Psychiatry & Psychotherapy Podcast. Emotion Connection. https://www.psychiatrypodcast.com/psychiatry-psychotherapy-podcast/2019/5/1/marijuana-and-mental-health?rq=marijuana
Puder, D. (Host). (2019, Oct. 23). Does Cannabis Use Increase Schizophrenia and Psychosis? (No. 64) [Audio podcast episode]. In Psychiatry & Psychotherapy Podcast. Emotion Connection. https://www.psychiatrypodcast.com/psychiatry-psychotherapy-podcast/2019/10/17/does-cannabis-increase-schizophrenia-and-psychosis-thc?rq=marijuana
Puder, D. (Host). (2024, January 19). Adverse Childhood Experiences and Their Lasting Impact on Health: A Comprehensive Guide (No. 203) [Audio podcast episode]. In Psychiatry & Psychotherapy Podcast. Emotion Connection. https://www.psychiatrypodcast.com/psychiatry-psychotherapy-podcast/episode-203-adverse-childhood-experiences-and-their-lasting-impact-on-health-a-comprehensive-guide?rq=203
Puder, D. (Host). (2024, February 2). Adverse Childhood Experiences Part 2: Measurement, Impact on Future Mental Health, Dissociation, and Timing of Trauma (No. 204) [Audio podcast episode]. In Psychiatry & Psychotherapy Podcast. Emotion Connection. https://www.psychiatrypodcast.com/psychiatry-psychotherapy-podcast/episode-204-adverse-childhood-experiences-part-2-measurement-impact-on-future-mental-health-dissociation-and-timing-of-trauma?rq=204
Puder, D. (Host). (2024, July 19). Adverse Childhood Experiences - HPA axis & Brain changes: cortisol, amygdala, hippocampus, cytokines, & epigenetics (Part 3 of ACE series)
(No. 217) [Audio podcast episode]. In Psychiatry & Psychotherapy Podcast. Emotion Connection. https://www.psychiatrypodcast.com/psychiatry-psychotherapy-podcast/episode-217-adverse-childhood-experiences-part-3-aces?rq=217
Rault, O., Romeo, B., Butlen-Ducuing, F., Rari, E., Benyamina, A., & Martelli, C. (2022). Impact of cannabis use and its cessation on the dosage and the efficacy of antipsychotic drugs in in- and outpatients with schizophrenia taking medication: A systematic review and meta-analysis. Journal of Psychiatric Research, 156, 713–721. https://doi.org/10.1016/j.jpsychires.2022.11.012
Renard, J., Szkudlarek, H. J., Kramar, C. P., Jobson, C. E. L., Moura, K., Rushlow, W. J., & Laviolette, S. R. (2017). Adolescent THC Exposure Causes Enduring Prefrontal Cortical Disruption of GABAergic Inhibition and Dysregulation of Sub-Cortical Dopamine Function. Scientific reports, 7(1), 11420. https://doi.org/10.1038/s41598-017-11645-8
Roberts, R. E., Roberts, C. R., & Xing, Y. (2010). One-year incidence of suicide attempts and associated risk and protective factors among adolescents. Archives of suicide research : official journal of the International Academy for Suicide Research, 14(1), 66–78. https://doi.org/10.1080/13811110903479078
Rodas, J. D., George, T. P., & Hassan, A. N. (2024). A Systematic Review of the Clinical Effects of Cannabis and Cannabinoids in Posttraumatic Stress Disorder Symptoms and Symptom Clusters. The Journal of clinical psychiatry, 85(1), 23r14862. https://doi.org/10.4088/JCP.23r14862
Rossi, G., & Beck, M. (2020). A little dab will do: a case of cannabis-induced psychosis. Cureus, 12(9). https://pmc.ncbi.nlm.nih.gov/articles/PMC7544610/
Ruglass, L. M., Shevorykin, A., Radoncic, V., Smith, K. M. Z., Smith, P. H., Galatzer-Levy, I. R., Papini, S., & Hien, D. A. (2017). Impact of Cannabis Use on Treatment Outcomes among Adults Receiving Cognitive-Behavioral Treatment for PTSD and Substance Use Disorders. Journal of Clinical Medicine, 6(2), 14. https://doi.org/10.3390/jcm6020014
Sabe, M., Zhao, N., & Kaiser, S. (2020). Cannabis, nicotine and the negative symptoms of schizophrenia: Systematic review and meta-analysis of observational studies. Neuroscience and biobehavioral reviews, 116, 415–425. https://doi.org/10.1016/j.neubiorev.2020.07.007
Schaefer, J. D., Hamdi, N. R., Malone, S. M., Vrieze, S., Wilson, S., McGue, M., & Iacono, W. G. (2021). Associations between adolescent cannabis use and young-adult functioning in three longitudinal twin studies. Proceedings of the National Academy of Sciences, 118(14), e2013180118. https://doi.org/10.1073/pnas.2013180118
Scheffler, F., Phahladira, L., Luckhoff, H., du Plessis, S., Asmal, L., Kilian, S., Di Forti, M., Murray, R., & Emsley, R. (2021). Cannabis use and clinical outcome in people with first-episode schizophrenia spectrum disorders over 24 months of treatment. Psychiatry Research, 302, 114022. https://doi.org/10.1016/j.psychres.2021.114022
Schoeler, T., Ferris, J. & Winstock, A.R. Rates and correlates of cannabis-associated psychotic symptoms in over 230,000 people who use cannabis. Transl Psychiatry 12, 369 (2022). https://doi.org/10.1038/s41398-022-02112-8
Schoeler, T., Baldwin, J. R., Martin, E., et al. (2024). Assessing rates and predictors of cannabis-associated psychotic symptoms across observational, experimental, and medical research. Nature Mental Health, 2(865–876). https://doi.org/10.1038/s44220-024-00261-x
Scott, J. C., Slomiak, S. T., Jones, J. D., Rosen, A. F. G., Moore, T. M., & Gur, R. C. (2018). Association of cannabis with cognitive functioning in adolescents and young adults: A systematic review and meta-analysis. JAMA Psychiatry, 75(6), 585–595. https://doi.org/10.1001/jamapsychiatry.2018.0335
Sherif, M., Radhakrishnan, R., D’Souza, D. C., & Ranganathan, M. (2016). Human laboratory studies on cannabinoids and psychosis. Biological Psychiatry, 79(7), 526–538. https://doi.org/10.1016/j.biopsych.2016.01.011
Shimonovich, M., Pearce, A., Thomson, H., Keyes, K., & Katikireddi, S. V. (2021). Assessing causality in epidemiology: Revisiting Bradford Hill to incorporate developments in causal thinking. European Journal of Epidemiology, 36, 873–887. https://doi.org/10.1007/s10654-020-00703-7
Silins, E., Horwood, L. J., Patton, G. C., Fergusson, D. M., Olsson, C. A., Hutchinson, D. M., Spry, E., Toumbourou, J. W., Degenhardt, L., Swift, W., Coffey, C., Tait, R. J., Letcher, P., Copeland, J., Mattick, R. P., & Cannabis Cohorts Research Consortium (2014). Young adult sequelae of adolescent cannabis use: an integrative analysis. The lancet. Psychiatry, 1(4), 286–293. https://doi.org/10.1016/S2215-0366(14)70307-4
Solowij, N., Broyd, S., Greenwood, L. M., van Hell, H., Martelozzo, D., Rueb, K., Todd, J., Liu, Z., Galettis, P., Martin, J., Murray, R., Jones, A., Michie, P. T., & Croft, R. (2019). A randomised controlled trial of vaporised Δ9-tetrahydrocannabinol and cannabidiol alone and in combination in frequent and infrequent cannabis users: acute intoxication effects. European archives of psychiatry and clinical neuroscience, 269(1), 17–35. https://doi.org/10.1007/s00406-019-00978-2
Sorkhou, M., Dent, E. L., & George, T. P. (2024). Cannabis use and mood disorders: a systematic review. Frontiers in public health, 12, 1346207. https://doi.org/10.3389/fpubh.2024.1346207
Spindle, T. R., Zamarripa, C. A., Russo, E., Pollak, L., Bigelow, G., Ward, A. M., Tompson, B., Sempio, C., Shokati, T., Klawitter, J., Christians, U., & Vandrey, R. (2024). Vaporized D-limonene selectively mitigates the acute anxiogenic effects of Δ9-tetrahydrocannabinol in healthy adults who intermittently use cannabis. Drug and Alcohol Dependence, 257, 111267. https://doi.org/10.1016/j.drugalcdep.2024.111267
Substance Abuse and Mental Health Services Administration (SAMHSA). (n.d.). Substance Abuse and Mental Health Services Administration. U.S. Department of Health and Human Services. Retrieved January 17, 2025, from https://www.samhsa.gov/
Substance Abuse and Mental Health Services Administration (SAMHSA). (2022a). 2021 National Survey on Drug Use and Health: National maps of prevalence estimates, by state. U.S. Department of Health and Human Services. https://www.samhsa.gov/data/sites/default/files/reports/rpt39463/2021NSDUHsaeMaps110122/2021NSDUHsaeMap110122.htm#fig2e
Substance Abuse and Mental Health Services Administration (SAMHSA). (2022b). Key substance use and mental health indicators in the United States: Results from the 2021 National Survey on Drug Use and Health (HHS Publication No. PEP22-07-01-005, NSDUH Series H-57). Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration. https://www.samhsa.gov/data/report/2021-nsduh-annual-national-report
U.S. Department of Veterans Affairs. (2023). Management of Posttraumatic Stress Disorder and Acute Stress Disorder: VA/DOD clinical practice guidelines. https://www.healthquality.va.gov/guidelines/MH/ptsd/
Van Green, T. (2022, November 22). Americans overwhelmingly say marijuana should be legal for medical or recreational use. Pew Research Center. https://www.pewresearch.org/short-reads/2022/11/22/americans-overwhelmingly-say-marijuana-should-be-legal-for-medical-or-recreational-use/
Vaucher, J., Keating, B. J., Lasserre, A. M., Gan, W., Lyall, D. M., Ward, J., Smith, D. J., Pell, J. P., Sattar, N., Paré, G., & Holmes, M. V. (2018). Cannabis use and risk of schizophrenia: A Mendelian randomization study. Molecular Psychiatry, 23(6), 1287–1292. https://doi.org/10.1038/mp.2016.252
Vaughn, S. E., Strawn, J. R., Poweleit, E. A., Sarangdhar, M., & Ramsey, L. B. (2021). The Impact of Marijuana on Antidepressant Treatment in Adolescents: Clinical and Pharmacologic Considerations. Journal of personalized medicine, 11(7), 615. https://doi.org/10.3390/jpm11070615
Verweij, K. J. H., Vink, J. M., Abdellaoui, A., Gillespie, N. A., Derks, E. M., & Treur, J. L. (2022). The genetic aetiology of cannabis use: From twin models to genome-wide association studies and beyond. Translational Psychiatry, 12(1), 489. https://doi.org/10.1038/s41398-022-02215-2
Vid Prkačin, M., Banovac, I., Petanjek, Z., & Hladnik, A. (2023). Cortical interneurons in schizophrenia - cause or effect?. Croatian medical journal, 64(2), 110–122. https://doi.org/10.3325/cmj.2023.64.110
von Wrede, R., Helmstaedter, C., & Surges, R. (2021). Cannabidiol in the Treatment of Epilepsy. Clinical drug investigation, 41(3), 211–220. https://doi.org/10.1007/s40261-021-01003-y
Wade, N. E., Wallace, A. L., Huestis, M. A., Lisdahl, K. M., Sullivan, R. M., & Tapert, S. F. (2024). Cannabis use and neurocognitive performance at 13–14 years old: Optimizing assessment with hair toxicology in the Adolescent Brain Cognitive Development (ABCD) study. Addictive Behaviors, 150, 107930. https://doi.org/10.1016/j.addbeh.2023.107930
Weeks, M., & Colman, I. (2017). Predictors of Suicidal Behaviors in Canadian Adolescents with No Recent History of Depression. Archives of suicide research : official journal of the International Academy for Suicide Research, 21(2), 354–364. https://doi.org/10.1080/13811118.2016.1193076
Weiland, B. J., Thayer, R. E., Depue, B. E., Sabbineni, A., Bryan, A. D., & Hutchison, K. E. (2015). Daily marijuana use is not associated with brain morphometric measures in adolescents or adults. The Journal of neuroscience : the official journal of the Society for Neuroscience, 35(4), 1505–1512. https://doi.org/10.1523/JNEUROSCI.2946-14.2015
Wilkinson, S. T., Stefanovics, E., & Rosenheck, R. A. (2015). Marijuana use is associated with worse outcomes in symptom severity and violent behavior in patients with posttraumatic stress disorder. The Journal of Clinical Psychiatry, 76(9), 1174–1180. https://doi.org/10.4088/JCP.14m09475
Xue, S., Husain, M. I., Zhao, H., & Ravindran, A. V. (2021). Cannabis Use and Prospective Long-Term Association with Anxiety: A Systematic Review and Meta-Analysis of Longitudinal Studies: Usage du cannabis et association prospective à long terme avec l'anxiété: une revue systématique et une méta-analyse d'études longitudinales. Canadian journal of psychiatry. Revue canadienne de psychiatrie, 66(2), 126–138. https://doi.org/10.1177/0706743720952251
Zamarripa, C. A., Spindle, T. R., Surujunarain, R., Weerts, E. M., Bansal, S., Unadkat, J. D., Paine, M. F., & Vandrey, R. (2023). Assessment of orally administered Δ9-tetrahydrocannabinol when coadministered with cannabidiol on Δ9-tetrahydrocannabinol pharmacokinetics and pharmacodynamics in healthy adults: A randomized clinical trial. JAMA Network Open, 6(2), e2254752. https://doi.org/10.1001/jamanetworkopen.2022.54752
Zammit, S., Allebeck, P., Andreasson, S., Lundberg, I., & Lewis, G. (2002). Self-reported cannabis use as a risk factor for schizophrenia in Swedish conscripts of 1969: Historical cohort study. BMJ, 325(7374), 1199. https://doi.org/10.1136/bmj.325.7374.1199
Zuardi, A. W., Teixeira, N. A., & Karniol, I. C. (1984). Pharmacological interaction of the effects of delta 9-trans-tetrahydrocannabinol and cannabidiol on serum corticosterone levels in rats. Archives internationales de pharmacodynamie et de therapie, 269(1), 12–19. https://pubmed.ncbi.nlm.nih.gov/6087750/